FUJIFILM Healthcare Americas Corporation has won the 2026 MedTech Breakthrough Award for “Best New Endoscopy Technology Solution” for its ELUXEO 8000 Endoscopic Imaging System in combination with the EG-840TP ultra slim therapeutic gastroscope. The recognition places Fujifilm’s latest endoscopy platform within a wider clinical and commercial shift toward advanced interventional upper gastrointestinal procedures, third space endoscopy and minimally invasive therapeutic care.
The award itself is not the most important part of the story. For clinicians, hospitals and medtech competitors, the larger signal is that endoscopy innovation is moving beyond incremental camera upgrades and into procedure-enabling ecosystems, where imaging, scope design, light modes, maneuverability and workflow efficiency need to work together inside increasingly complex anatomy.
Why Fujifilm’s ELUXEO 8000 award matters beyond another medtech recognition cycle
Fujifilm’s second consecutive win in the MedTech Breakthrough endoscopy category gives the medical device manufacturer a useful credibility marker in a crowded imaging market, but its strategic relevance lies in the clinical workflows the platform is targeting. Interventional upper gastrointestinal endoscopy and third space endoscopy are technically demanding procedures that depend heavily on stable visualization, fine access, tissue-layer recognition and bleeding control. A system that can make those procedures more predictable has potential value beyond marketing differentiation.
That matters because hospitals are increasingly looking for technologies that support minimally invasive interventions while reducing procedural burden and recovery time. Endoscopy has already moved from a diagnostic specialty into a therapeutic platform across gastrointestinal disease, but advanced procedures remain dependent on operator experience and equipment quality. Fujifilm’s positioning suggests that the next phase of competition will not be decided only by image sharpness, but by whether imaging systems can help clinicians navigate procedural risk in real time.
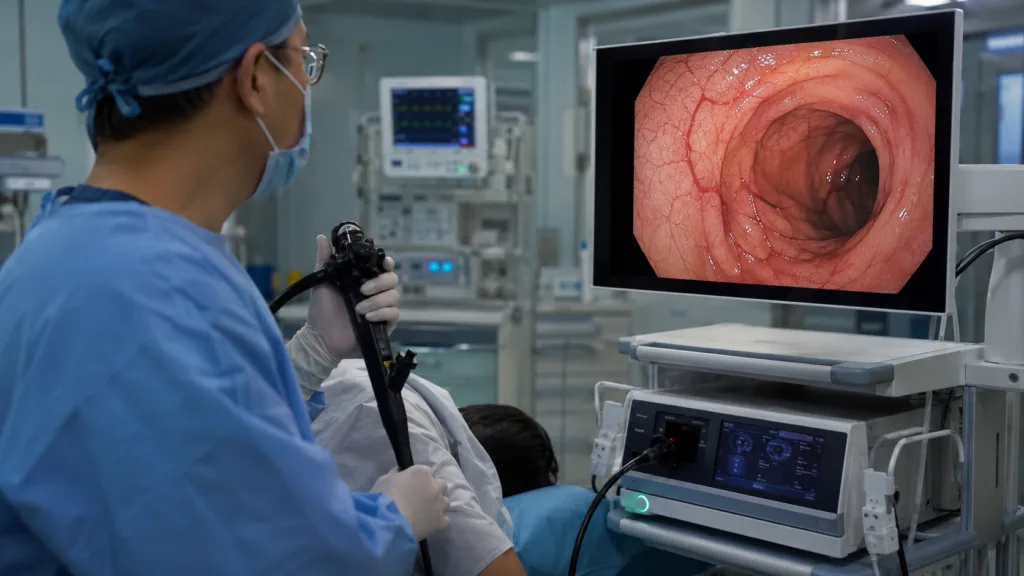
The limitation is that award recognition does not automatically translate into hospital adoption. Procurement decisions in endoscopy are shaped by installed base, physician preference, training requirements, service support, compatibility with existing towers and capital expenditure cycles. Fujifilm now has stronger external validation for ELUXEO 8000, but the harder commercial test is whether the platform can convert specialist enthusiasm into broader institutional purchasing.
How ELUXEO 8000 and EG-840TP address the real difficulty of third space endoscopy
Third space endoscopy has become one of the more technically sophisticated areas of gastrointestinal intervention because it requires clinicians to work within the submucosal layer rather than simply inspect the mucosal surface. In practical terms, that means visibility, tissue differentiation and access are not optional enhancements. They are central to whether the procedure can be performed safely and efficiently.
The ELUXEO 8000 system’s Amber-Red Color Imaging mode is positioned around that need. By improving visualization of blood vessels and tissue layers in the submucosal space, the technology directly targets one of the major challenges in third space procedures: recognizing and managing structures that may be difficult to distinguish under standard white-light imaging. If the mode helps clinicians identify bleeding sources or critical tissue planes more clearly, it could support procedural confidence in cases where visibility can change quickly.
However, the clinical value of any enhanced imaging mode depends on real-world usability. Hospitals and clinicians will likely watch whether Amber-Red Color Imaging delivers consistent benefit across different patient anatomies, bleeding conditions, operator skill levels and procedure types. Enhanced visualization can be powerful, but adoption usually accelerates only when specialists believe it improves decision-making without adding complexity to an already demanding procedure.
Why ultra slim therapeutic gastroscopes are becoming more important in advanced GI care
The EG-840TP gastroscope is significant because it addresses access, not just imaging. Fujifilm has paired the ELUXEO 8000 with an ultra slim therapeutic gastroscope designed for narrow lumens, strictures and submucosal tunneling. That combination matters because many advanced endoscopic procedures fail not because the image is poor, but because the device cannot move comfortably through difficult anatomy while maintaining therapeutic capability.
The 7.9 millimeter outer diameter, enhanced angulation, water jet function and CMOS imaging give the scope a set of practical features aimed at specialists performing complex upper gastrointestinal work. In procedures where maintaining visibility is difficult, a water jet function can be more than a convenience. It can help preserve the visual field, especially when blood, debris or fluid interrupts the procedure.
The commercial question is whether this pairing becomes a specialist-only tool or part of a broader endoscopy platform strategy. Ultra slim therapeutic scopes may appeal strongly to centers already performing advanced third space endoscopy, but broader adoption will depend on case volume, training pathways and whether the device can justify its role across multiple procedure types. For medtech manufacturers, the most attractive products are often those that solve a specialist problem while still fitting into wider hospital workflows.
What this reveals about competition in endoscopic imaging platforms
Fujifilm’s recognition highlights a broader competitive shift in endoscopy, where manufacturers are trying to move from single-device upgrades to integrated procedural ecosystems. Endoscopy towers, light modes, processors, scopes and accessories increasingly need to be assessed as a connected platform rather than separate products. That shift favors companies that can align imaging science with procedural ergonomics and workflow efficiency.
For Fujifilm, the ELUXEO 8000 and EG-840TP combination supports a message that the company is not merely competing on visualization, but on enabling emerging therapeutic techniques. That distinction is important because hospitals may be more willing to invest in capital equipment when the system is tied to expanded procedural capability, not just better pictures. If a platform supports new services or helps retain complex cases in-house, its value proposition becomes more strategic.
Still, Fujifilm faces the same challenge as every advanced medtech platform supplier: demonstrating that the technology changes outcomes, efficiency or economics in ways that matter to buyers. A stronger image or more maneuverable scope is clinically appealing, but hospital administrators often need evidence that the investment improves throughput, reduces referrals, supports reimbursement opportunities or strengthens service-line differentiation.
Why the MedTech Breakthrough win could support Fujifilm’s positioning with hospitals
The MedTech Breakthrough Award gives Fujifilm a third-party recognition point at a time when endoscopy departments are evaluating technology refresh cycles, procedural expansion and competitive differentiation. Awards do not replace clinical evidence, but they can help reinforce confidence among procurement committees and specialist teams comparing multiple vendors.
The recognition may be especially useful because Fujifilm won the same category in 2025 for its EN-840T Double Balloon Enteroscopy Scope. Consecutive recognition suggests continuity in product development rather than a one-off device launch. For hospitals, that can matter because endoscopy investments are long-cycle decisions. Institutions are not only buying equipment, they are committing to a vendor relationship involving training, maintenance, upgrades and procedural support.
The risk is that external recognition can create visibility without resolving adoption friction. Capital equipment purchases remain sensitive to budgets, reimbursement pressures and competing priorities across hospital systems. Even when clinicians value new technology, hospitals may delay purchases if the clinical service line cannot prove enough near-term financial or operational impact.
What clinicians and medtech observers will watch next in advanced Fujifilm endoscopy
The next phase for Fujifilm will likely depend on how ELUXEO 8000 performs in specialist practice and whether its feature set becomes associated with meaningful procedural advantages. Clinicians tracking advanced endoscopy will want to see whether Amber-Red Color Imaging becomes a practical aid in third space procedures, whether the EG-840TP gastroscope improves access in difficult anatomy, and whether the combined platform can support more consistent outcomes across operator settings.
Industry observers will also watch whether Fujifilm can use the award to strengthen its competitive standing in the U.S. endoscopy market. Endoscopy remains a field where physician loyalty, installed equipment, training history and service reliability carry substantial weight. Fujifilm’s technology story is becoming sharper, but commercial expansion will require sustained execution beyond product recognition.
The broader implication is that advanced endoscopy is moving toward a more procedure-specific era. The winners will not simply be companies with brighter images or thinner scopes. They will be the manufacturers that help clinicians perform more complex interventions with better control, clearer visualization and fewer workflow compromises. Fujifilm’s latest award suggests it wants ELUXEO 8000 and EG-840TP to sit directly in that conversation.