PharmaJet has announced that its Tropis Intradermal Needle-free Injection System will be used to deliver about 1.4 million fractional inactivated polio vaccine doses during immunisation campaigns in Nigeria. The deployment, focused on children under five in Sokoto, comes as Nigeria remains free of wild poliovirus but continues to face circulating vaccine-derived poliovirus type 2 outbreaks.
Why PharmaJet’s Nigeria deployment matters for polio eradication strategy
The significance of the PharmaJet Tropis deployment lies less in the headline number of vaccine doses and more in what the campaign reveals about the next phase of polio eradication. Nigeria is no longer fighting wild poliovirus, which is a major public health achievement. The harder challenge now is stopping circulating vaccine-derived poliovirus type 2, a problem that tends to emerge where immunisation gaps allow weakened vaccine virus strains to circulate and mutate over time.
That makes vaccine delivery performance a strategic issue, not just an operational detail. In settings where coverage is uneven, the difference between a standard campaign and a campaign that reaches more children faster can have direct implications for outbreak control. Needle-free intradermal delivery of fractional-dose inactivated polio vaccine offers a practical route to stretch vaccine supply, reduce per-child cost, and simplify administration in field conditions. For Nigeria, that matters because polio eradication now depends on reaching difficult geographies, mobile populations, and communities where conventional immunisation workflows may struggle.
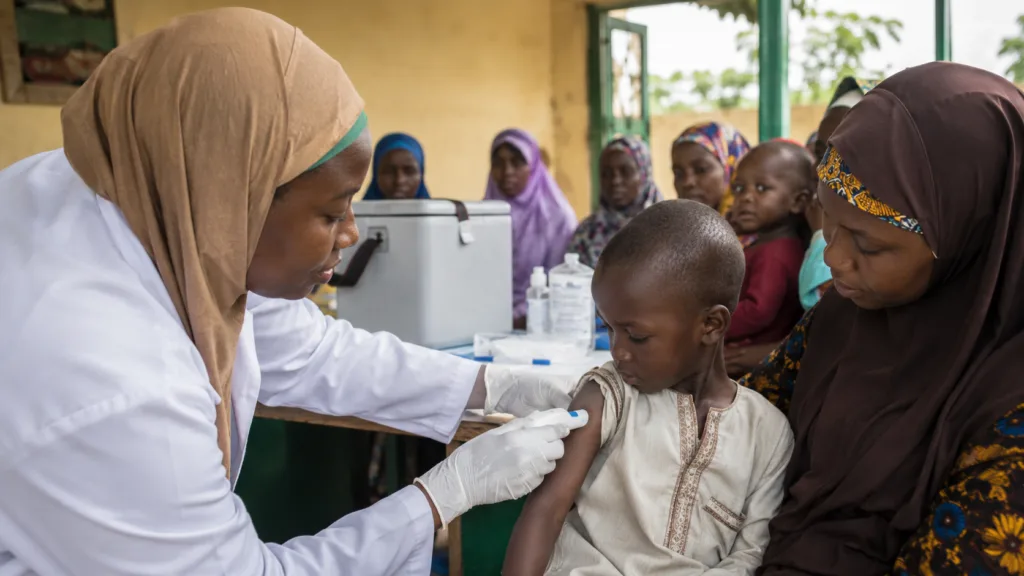
The commercial and public health question is whether this model can move from selective campaign use to a more routine role in immunisation systems. PharmaJet’s Tropis system has already been used in large-scale polio work across Pakistan, Afghanistan, Somalia, and Nigeria. That history gives the platform credibility, but the Nigeria campaign will still be watched for execution quality, training consistency, dose accuracy, and whether reported improvements in acceptability translate into measurable gains in field coverage.
How needle-free fractional-dose IPV changes the economics of vaccine delivery
The core advantage of the Tropis system is its fit with fractional-dose inactivated polio vaccine delivery. Intradermal administration allows a smaller vaccine quantity to be used per child, which can increase the number of children reached from available vaccine supply. In an eradication campaign, that is not a minor efficiency gain. It can change campaign planning, stock allocation, cold-chain burden, and the economics of repeated supplementary immunisation activities.
The reported cost reduction from earlier Nigerian routine immunisation research is especially important because polio eradication is increasingly a funding discipline as much as a scientific one. Global eradication programmes must sustain surveillance, rapid response, routine immunisation reinforcement, and outbreak campaigns even after wild poliovirus has disappeared from many regions. Any delivery model that can reduce total immunisation cost while improving coverage will attract attention from ministries of health, donor agencies, and implementation partners.
However, cost savings do not automatically guarantee system-wide adoption. Needle-free systems require device procurement, cartridge supply, training, maintenance planning, waste-management alignment, and confidence among vaccinators. The economics only work if higher coverage, faster administration, and lower dose costs outweigh the additional device-related logistics. For PharmaJet, the Nigeria deployment strengthens the evidence base, but the harder commercial question is whether ministries and implementing agencies will treat the device as a campaign tool or as part of a broader immunisation infrastructure shift.
Why Sokoto is a meaningful test case for needle-free polio campaigns
Sokoto is not just another campaign location. Northwestern Nigeria has been central to the country’s immunisation challenge because of hard-to-reach communities, security pressures, cross-border mobility, and the need for sustained local engagement. A campaign in this environment tests more than device performance. It tests whether a technology can function under real-world public health constraints where workforce capacity, community trust, and logistics all matter.
The use of master trainers across several Local Government Areas indicates that implementation quality is a key part of the strategy. Needle-free technology can improve speed and acceptability only if vaccinators are properly trained and confident. Poor technique, inconsistent dosing, or weak supervision could dilute the benefits that the platform is intended to deliver. That is why training infrastructure may be as important as the device itself.
For clinicians and public health officials, the appeal of needle-free delivery also extends to caregiver perception. In paediatric campaigns, fear of needles can reduce acceptance, slow administration, and create friction during house-to-house or community-based vaccination drives. A needle-free option may reduce that barrier, particularly when campaigns involve very young children. Still, acceptance must be proven locally. Community response in Sokoto will matter because public health technology succeeds only when it works culturally as well as technically.
What PharmaJet gains from wider Tropis adoption in global immunisation
For PharmaJet, the Nigeria campaign deepens Tropis’ positioning as an enabling platform in global immunisation rather than a niche device. The medical device manufacturer is not selling a therapeutic product in the conventional sense. It is selling a delivery system that can make vaccines easier, cheaper, or more acceptable to administer. That distinction matters because platform adoption depends on partnerships with governments, non-profit organisations, public health agencies, and vaccine manufacturers.
The public health pathway can be commercially attractive but slow. Global immunisation procurement is evidence-driven, cost-sensitive, and heavily influenced by implementation data. The strongest signal from the Nigeria announcement is that Tropis is being used after prior study work involving major immunisation stakeholders. That suggests the platform is moving through the kind of evidence-to-adoption cycle that matters in public-sector vaccine delivery.
The limitation is that campaign use does not necessarily translate into predictable recurring revenue. Supplementary immunisation activities are episodic, donor-dependent, and shaped by outbreak dynamics. PharmaJet’s longer-term opportunity will depend on whether needle-free intradermal delivery becomes embedded in routine immunisation schedules, emergency response campaigns, or future vaccine programmes beyond polio. The more diseases and vaccine types that can be linked to needle-free delivery benefits, the stronger the platform’s strategic value becomes.
Why this is not just a device story but a vaccine access story
The broader importance of the Tropis deployment is that vaccine innovation increasingly includes delivery innovation. The world often focuses on antigen design, mRNA platforms, viral vectors, and next-generation adjuvants. However, eradication campaigns can fail at the last mile even when effective vaccines exist. The operational gap between vaccine availability and vaccine administration remains one of the most persistent barriers in public health.
Needle-free systems address part of that last-mile problem by simplifying the interface between healthcare worker, child, and vaccine. In resource-limited or austere settings, reducing complexity can matter as much as improving scientific sophistication. If non-traditional healthcare workers can use the system effectively in house-to-house strategies, the technology could help expand campaign reach without relying solely on highly trained clinical personnel.
Yet the risk is that delivery technologies are sometimes overestimated when the underlying barriers are social, political, or infrastructural. In places where vaccine hesitancy, insecurity, weak surveillance, or poor campaign microplanning are the main constraints, a better delivery device may help but will not solve the problem alone. The strongest use case for Tropis will be where device-level efficiencies align with strong local mobilisation, credible health messaging, and reliable campaign execution.
What clinicians, regulators, and public health agencies will watch next
The next phase of scrutiny will likely focus on measurable campaign outcomes. Coverage improvement, administration speed, dose wastage, adverse event monitoring, vaccinator preference, caregiver acceptance, and campaign cost will all matter. Public health agencies will want to know whether the results seen in earlier studies are reproduced at larger scale and under campaign conditions.
Regulatory and procurement stakeholders will also watch the evidence threshold for broader adoption. The Tropis system has relevant global immunisation credentials, but each additional use case strengthens or weakens confidence depending on field performance. For a delivery device, the key questions are practical. Can it be trained quickly? Can it be supplied reliably? Can it reduce cost without introducing new operational bottlenecks? Can it maintain consistent administration quality across thousands of vaccinators and multiple local settings?
For Nigeria, the larger goal remains interruption of all poliovirus transmission by 2030. The PharmaJet deployment will not determine that outcome by itself, but it could contribute to the toolkit needed to close remaining immunisation gaps. For PharmaJet, the opportunity is to show that needle-free delivery can do more than improve user experience. It must prove that it can materially improve campaign performance in the environments where eradication is hardest.
Why the Tropis campaign could influence future vaccine delivery models
If the Nigeria campaign performs well, the implications could extend beyond polio. Many vaccine programmes face the same constraints that make polio eradication difficult: limited workforce capacity, cost pressure, cold-chain complexity, caregiver hesitancy, and the need to reach children outside conventional clinic settings. A proven needle-free intradermal model could therefore become relevant to future outbreak response and routine immunisation strategies.
That does not mean needle-free injection will replace conventional needle-and-syringe delivery across the board. The conventional model is inexpensive, familiar, and deeply embedded in health systems. PharmaJet’s strongest argument is likely not replacement, but targeted use where fractional dosing, speed, acceptability, or access challenges create a clear advantage.
The Nigeria deployment is therefore best understood as a pressure test for a more flexible vaccine delivery future. If Tropis can help deliver 1.4 million fractional inactivated polio vaccine doses efficiently in Sokoto, it will strengthen the case for delivery systems as a serious lever in global health strategy. The bigger story is not simply that a needle-free device is being used in a polio campaign. It is that the final mile of vaccine delivery is becoming a field of innovation in its own right.