Why Boehringer’s TL1A/IL23 bispecific bet could reshape IBD drug development

Find out why Boehringer is betting big on Simcere’s bispecific SIM0709 to challenge the IBD treatment ceiling.

Find out why Boehringer is betting big on Simcere’s bispecific SIM0709 to challenge the IBD treatment ceiling.

Can TPC-026 fill the gap after GLP-1s? Find out how TETRAPHARM’s novel compound could reshape long-term metabolic care.

Hanmi taps Sanfer to launch GLP-1 drug efpeglenatide in Mexico. Find out what this deal reveals about Latin America’s obesity drug market strategy.

Find out how Fujirebio’s new Lumipulse pTau 217 CSF assay could shape the future of Alzheimer’s biomarker research and platform-based diagnostics.

Find out how USDTL’s integration with CourtFact could reshape digital drug testing workflows for justice-involved programs.

Skyhawk’s SKY-0515 shows functional gains in Huntington’s disease. Find out how RNA modulation could alter the treatment landscape.
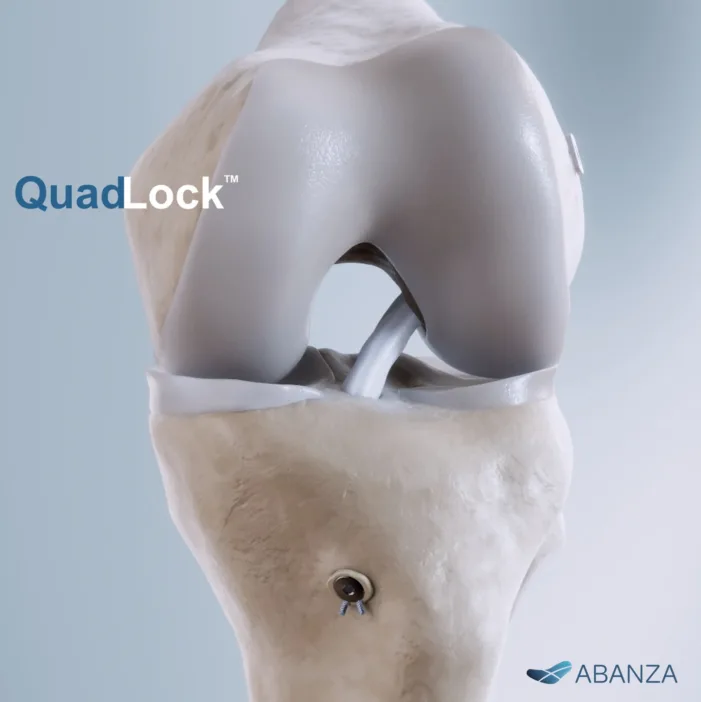
Abanza Inc. has received U.S. Food and Drug Administration (FDA) 510(k) clearance for QuadLock, its novel knotless fixation system for anterior cruciate ligament (ACL) reconstruction. The system, which features a bidirectional tension-adjustable design, reportedly offers over a 500% reduction in cyclic displacement compared to conventional fixation methods such as cortical buttons and interference screws, targeting […]

Genentech’s CT-388 hit 22.5% weight loss in Phase II. Find out what this means for obesity drugs, clinical durability, and market strategy in 2026.

Mirum adds brelovitug in HDV through Bluejay deal. See how this could redefine rare liver disease leadership ahead of AZURE Phase 3 results in 2026.

Fortitude Biomedicines launches with $13M to develop glue-payload ADCs for autoimmune and cancer treatment. Find out what makes this platform different.