Why the SRS–Lenke–Aubin 3D classification could reshape adolescent scoliosis research

The SRS–Lenke–Aubin 3D classification brings new depth to AIS care. Find out how it could reshape surgery, planning, and clinical trial design.

The SRS–Lenke–Aubin 3D classification brings new depth to AIS care. Find out how it could reshape surgery, planning, and clinical trial design.
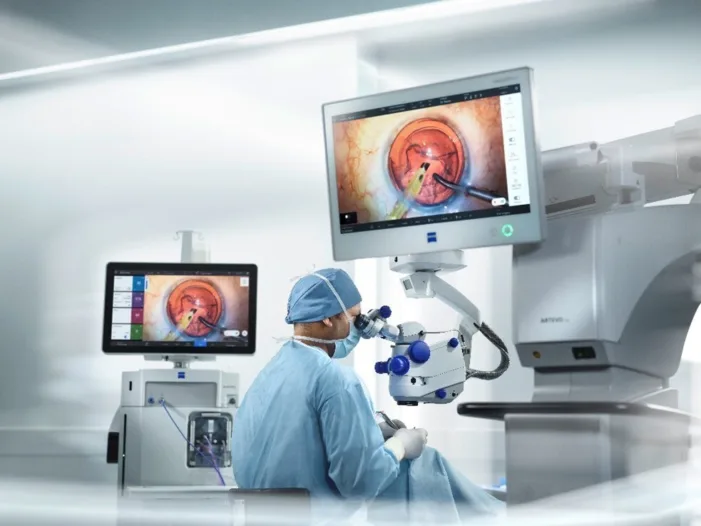
ZEISS receives NMPA approval in China for ARTEVO 750 and 850 ophthalmic microscopes. Find out how these devices are shaping the future of eye surgery.

FUGO Precision 3D is debuting its centrifugal 3D printing tech at LMT LAB DAY 2026. Find out how this could transform dental manufacturing workflows.

CREATE Medicines shows redosable B cell depletion in primates with in vivo CAR-T platform. Find out what this means for autoimmune and oncology therapy.
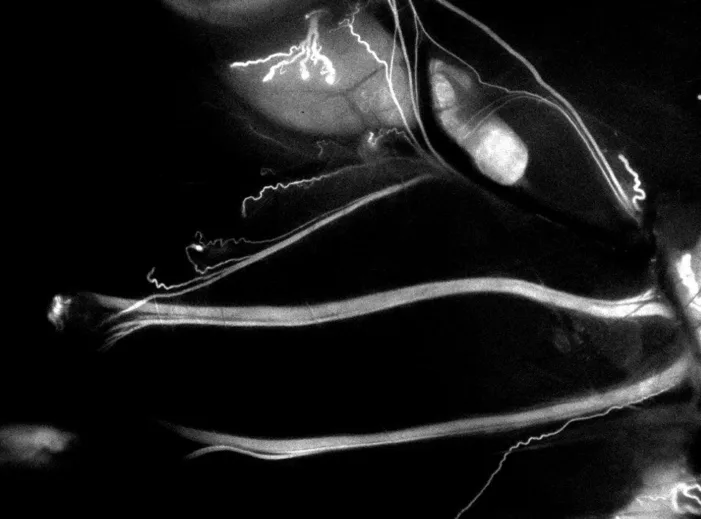
Trace Biosciences receives IND approval for LGW16-03. Find out what this means for nerve imaging and the future of surgical precision.

Find out how Cynosure Lutronic’s Mosaic 3D laser device could redefine non-ablative resurfacing after CE mark approval under EU MDR.

Frontage Laboratories expands US and China early phase clinical sites with radiolabel, BE, and oncology focus. Find out what this means for CRDMOs in 2026.

ZTALMY gains commercial launch in China for CDKL5 seizures. Find out what this means for rare disease drug access and neurology strategy.

Eli Lilly and Company has announced a $3.5 billion investment to build a new injectable medicine and device manufacturing facility in Lehigh Valley, Pennsylvania. The site will focus on producing next-generation weight-loss medicines, including the investigational triple hormone receptor agonist retatrutide. Set to become operational by 2031, this is Lilly’s fourth new U.S. manufacturing site […]

Find out how UVDI’s V-MIX UV-C disinfection system is pushing upper-room air safety beyond hospitals into schools, offices, and airports.