NiKang Therapeutics unveils NKT5097 as dual CDK2/4 degrader for solid tumor strategy

NiKang Therapeutics’ NKT5097 enters Phase 1 as a dual CDK2/4 degrader aiming to overcome breast cancer resistance. Explore what this strategy changes.

NiKang Therapeutics’ NKT5097 enters Phase 1 as a dual CDK2/4 degrader aiming to overcome breast cancer resistance. Explore what this strategy changes.

FDA clears Prilenia’s PREVAiLS ALS trial. Can pridopidine validate neuroprotection and shift the treatment paradigm? Read the full analysis here.

IDEAYA Biosciences files IND for IDE574, a KAT6/7 inhibitor for breast and lung cancer. Learn how this new class may change epigenetic treatment strategies.

Can four doses replace chronic ITP therapy? Find out how Novartis’ ianalumab performed in the VAYHIT2 trial and what it could mean for autoimmune care.

TECVAYLI and DARZALEX FASPRO show major survival benefits in second-line multiple myeloma. Find out what the MajesTEC-3 trial means for clinical practice.

ProVerum wins FDA approval for ProVee, a permanent urethral stent for BPH offering fast recovery and no catheter. See how it could disrupt the urology device market.

Eli Lilly and Indiana University are launching a $40M clinical trial and Alzheimer’s research initiative in Indiana. See what it could mean for U.S. pharma.

Philips unveils AVW 16 and new AI integrations at RSNA 2025. Find out how its cloud-native platform strategy could reshape diagnostic imaging workflows.
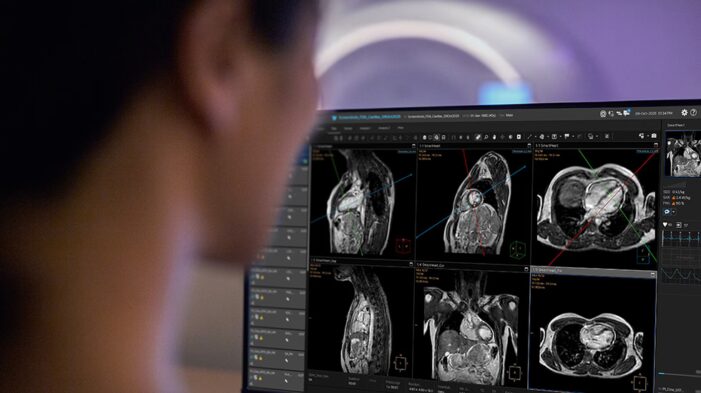
Find out how Philips’ new AI-powered cardiac MR innovations could transform access, speed, and diagnostic precision in cardiovascular imaging.