Cynosure Lutronic sets stage for global Mosaic 3D rollout with IMCAS 2026 momentum

Find out how Cynosure Lutronic’s Mosaic 3D laser device could redefine non-ablative resurfacing after CE mark approval under EU MDR.

Find out how Cynosure Lutronic’s Mosaic 3D laser device could redefine non-ablative resurfacing after CE mark approval under EU MDR.

Find out how UVDI’s V-MIX UV-C disinfection system is pushing upper-room air safety beyond hospitals into schools, offices, and airports.

KORU Medical’s FreedomEDGE gets FDA clearance for RYSTIGGO delivery in clinics. Find out how this expands access to gMG therapy and reshapes infusion workflows.

Demeetra’s acquisition of Hera BioLabs consolidates Super piggyBac IP. Find out how this could reshape gene therapy tools and licensing frameworks.

Find out how Fujirebio’s new Lumipulse pTau 217 CSF assay could shape the future of Alzheimer’s biomarker research and platform-based diagnostics.

Find out how USDTL’s integration with CourtFact could reshape digital drug testing workflows for justice-involved programs.
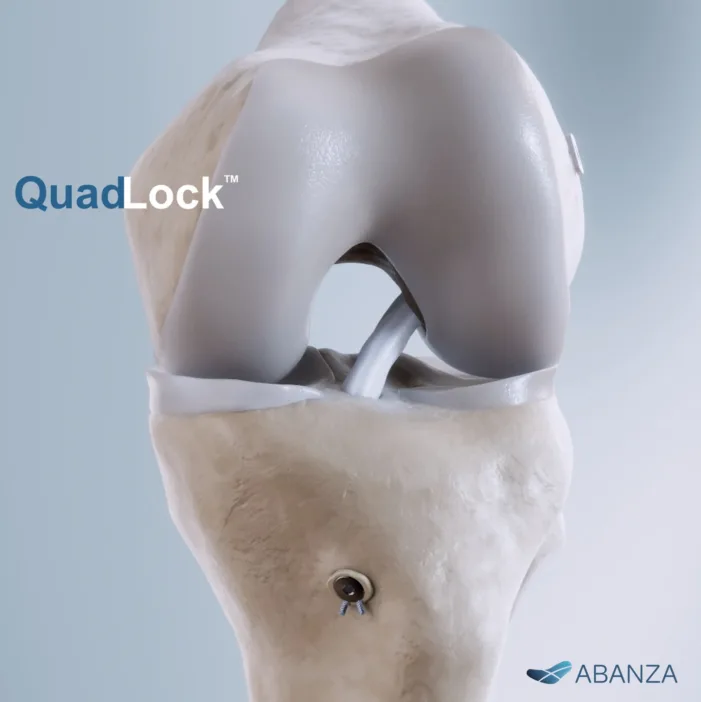
Abanza Inc. has received U.S. Food and Drug Administration (FDA) 510(k) clearance for QuadLock, its novel knotless fixation system for anterior cruciate ligament (ACL) reconstruction. The system, which features a bidirectional tension-adjustable design, reportedly offers over a 500% reduction in cyclic displacement compared to conventional fixation methods such as cortical buttons and interference screws, targeting […]

Merz Therapeutics files EMA application to expand XEOMIN for pediatric spasticity. Find out how this could shift the treatment landscape for cerebral palsy.

Aesyra’s AesyBite Active reduced sleep bruxism by over 60% in trial. Find out what this could mean for digital therapeutics and FDA approval in 2026.

Vitaline rebrands from Dript IV Therapy to signal its shift toward clinical-grade hydration services in senior care. Find out what this change means now.