Why Aphranel’s EU MDR approval could sharpen competition in CaHA dermal fillers

Aphranel’s CaHA filler has secured EU MDR certification. Find out what this means for regenerative aesthetics and Europe’s filler market.

Aphranel’s CaHA filler has secured EU MDR certification. Find out what this means for regenerative aesthetics and Europe’s filler market.

Tempus AI expands Lens with agentic AI for oncology drug development. Find out how it could reshape trials, evidence generation and biopharma R&D.

Can Veracyte’s Decipher Prostate test change chemotherapy decisions in metastatic prostate cancer? Find out what ENZAMET data means today.
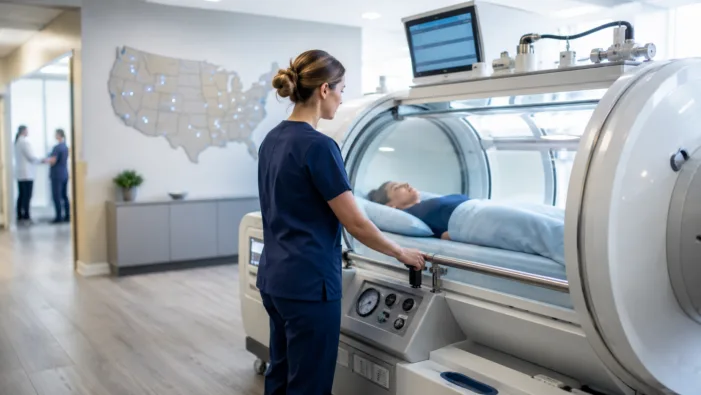
HBOT demand is rising beyond hospitals. MD Hyperbaric’s clinic expansion tests whether recovery care can scale without losing clinical rigor.
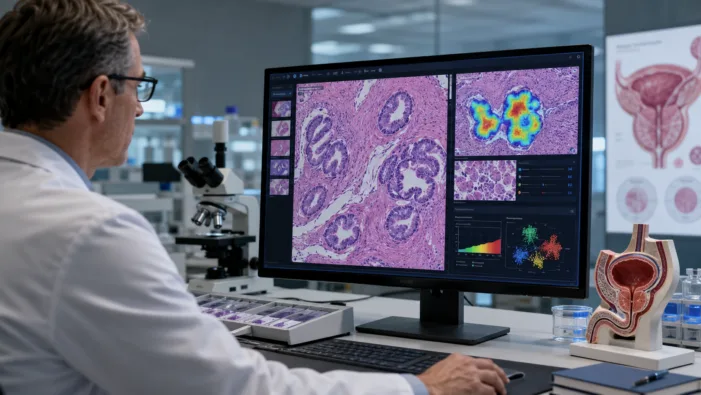
Acupath adds ArteraAI Prostate Test to its pathology workflow. Find out what it means for AI prostate cancer risk stratification today.

Coredio’s AI heart failure platform may bring hemodynamic insight into home care. See why FDA breakthrough status matters now.
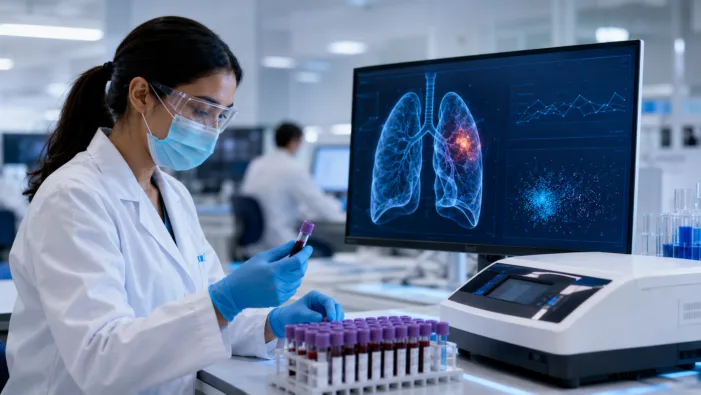
DELFI Diagnostics can now offer FirstLook Lung in all 50 states. Find out what this means for blood-based cancer screening.

Find out why Seer’s activist battle could reshape investor thinking on proteomics, diagnostics platforms and biotech value creation.

Hologic’s Breast Cancer Index is changing endocrine therapy decisions. The bigger test is adoption in real-world breast cancer care.

DePuy Synthes wants to remove a robotics bottleneck. Its Gemtrack deal could change joint surgery navigation if regulators agree.