Mitria Medical, the Cleveland-based structural heart company spun out of Cleveland Clinic Innovations in 2019, announced the first implantation of its Subvalvular Spacer in the United States, performed at Cleveland Clinic under the FDA’s Single Patient Expanded Access programme for a patient with severe functional mitral regurgitation. The procedure marks the device’s regulatory debut on U.S. soil after earlier cases in Uzbekistan and Paraguay, and brings a distinctly different mechanical philosophy to a field dominated by leaflet-clip technology.
Why anatomy-specific design matters more than market share in functional mitral regurgitation
The clinical significance of the Subvalvular Spacer lies not in what it does broadly, but in what it does differently. Functional mitral regurgitation arises not from primary valve disease but from left ventricular dysfunction and geometric distortion, causing the leaflets to be pulled away from each other during systole. Established transcatheter options, primarily edge-to-edge repair devices, address this by mechanically clipping the leaflets together. That approach works well for a defined subset of patients, but it carries anatomical constraints. When the posterior leaflet is significantly tethered or the annular geometry makes adequate leaflet coaptation difficult to achieve by clipping alone, currently available percutaneous options often become unsuitable or sub-optimal.
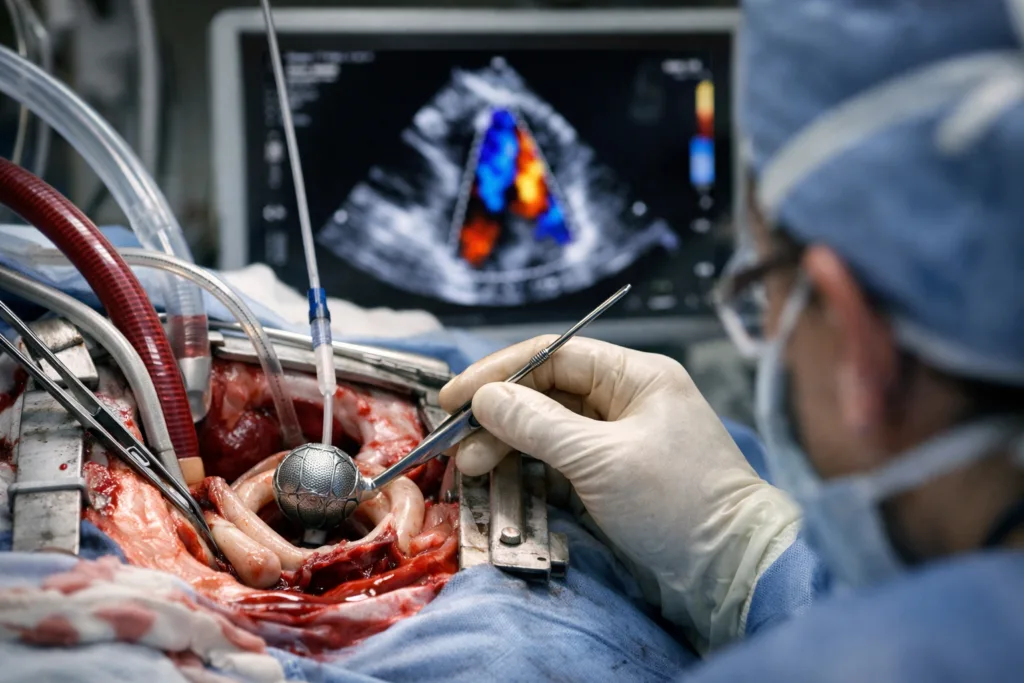
The Subvalvular Spacer is designed to intervene at a different point in that pathological chain. Rather than forcing leaflet contact, the nitinol braided device sits at the hinge point of the posterior leaflet, lifting and supporting it from below to restore coaptation while leaving anterior leaflet motion largely undisturbed. The claim that it preserves natural valve orifice area is clinically meaningful: procedures that create a double-orifice valve, as edge-to-edge repair does, can restrict transmitral flow, particularly in patients with already-compromised ventricular function. Whether the Subvalvular Spacer’s subvalvular position genuinely avoids those haemodynamic trade-offs at scale is a question that only a properly powered clinical trial can answer.
What the compassionate use pathway does and does not tell investors and clinicians
Single patient expanded access is not a clinical trial and should not be read as one. The FDA grants compassionate use when a patient faces a serious or life-threatening condition and no satisfactory alternative exists, not because a device has met a pre-specified evidence threshold. The pathway serves individual patients, not regulatory programmes. That said, the regulatory decision to permit the procedure, and the choice of Cleveland Clinic as the site, are signals worth noting. The FDA’s willingness to grant expanded access implies a minimum safety credibility review, and Cleveland Clinic’s institutional involvement brings procedural rigour and outcome reporting infrastructure that informal case series elsewhere might lack.
The three prior international cases, presented at the Transcatheter Cardiovascular Therapeutics conference, the American Association for Thoracic Surgery’s Mitral Conclave, and Miami Valves, suggest that Mitria Medical has been building a deliberate pre-pivotal dataset rather than accumulating cases opportunistically. That conference-driven disclosure strategy is consistent with a company preparing for an investigational device exemption application, the U.S. regulatory pathway required before a formal pivotal trial can begin. Clinicians tracking the space will note that the company has not yet announced an IDE submission, meaning there is likely at least one further regulatory step before a controlled U.S. trial begins enrolling.
How the Subvalvular Spacer fits against the competitive landscape for transcatheter mitral repair
The mitral transcatheter market remains structurally dominated by Abbott’s MitraClip and, more recently, Edwards Lifesciences’ PASCAL system, both of which have substantial clinical evidence bases and established reimbursement pathways. Both are edge-to-edge repair platforms, and both have been shown in landmark trials to reduce hospitalisation and improve quality of life in carefully selected patients. The Subvalvular Spacer is not, at this stage, competing with those platforms for the same patient population. Its stated focus on patients who are ineligible for existing therapies due to anatomy positions it initially as a complement rather than a substitute.
That positioning is strategically sensible for a privately held company without the commercial infrastructure to mount a head-to-head trial, but it also limits the size of the immediately addressable patient pool. Industry observers note that the real commercial opportunity depends on whether the device can eventually demonstrate outcomes in a broader functional mitral regurgitation population or whether its benefit remains confined to anatomically complex cases that current platforms cannot reach. The former would represent a significant market entry; the latter would position the device as a niche but potentially valuable addition to the mitral repair toolkit.
The Cleveland Clinic co-inventor structure adds a layer of institutional credibility but also introduces conflict-of-interest considerations that will need transparent management as the device moves toward formal trial phases. Both Jose Navia and Samir Kapadia hold equity ownership as co-inventors, and their institutional statements accompanying this announcement reflect that dual role. Regulatory watchers suggest that co-inventor involvement in early implantation cases is not unusual in structural heart innovation, but it underscores the importance of independent data monitoring committees and site diversity in any forthcoming pivotal study.
What the regulatory pathway forward actually requires
Functional mitral regurgitation has a complicated regulatory history in the United States. The COAPT trial, which underpinned the MitraClip indication, set a high bar for patient selection rigour, and the FDA has signalled repeatedly that it expects transcatheter mitral programmes to demonstrate net clinical benefit rather than procedural feasibility alone. For the Subvalvular Spacer to progress toward PMA approval, Mitria Medical will need to define a patient population clearly enough to power a pivotal trial, establish a primary endpoint acceptable to the FDA, and demonstrate durability data beyond the acute procedural results that compassionate use cases can provide.
The device’s nitinol braid construction and subvalvular deployment approach will also require detailed fatigue and durability characterisation, given the mechanical stress environment of a functioning mitral valve over years of cardiac cycles. Industry observers note that materials data from the international cases may partially address this, but a formal IDE submission will require comprehensive bench testing and, in all likelihood, a feasibility study with structured follow-up before pivotal enrolment begins.
What clinicians, regulators, and the structural heart community will watch next
The metrics that will define this device’s trajectory are largely prospective rather than retrospective. The field will want to see formal outcomes data from the international cases presented in peer-reviewed form rather than conference abstracts, a defined patient selection algorithm that can guide site screening consistency, and a clear timeline for IDE submission. Whether Cleveland Clinic will serve as a co-primary site in a formal trial, or whether Mitria Medical broadens its site network materially before pivotal enrolment, will also influence how quickly robust data accrues.
For the broader structural heart community, the Subvalvular Spacer represents a genuine design innovation that, if validated, could expand the treatable population for transcatheter mitral repair. The current announcement establishes U.S. procedural feasibility and institutional partnership. It does not yet establish clinical evidence. The distance between those two things, and how quickly Mitria Medical closes it, is what the market will be watching.