Bebird, the consumer-focused smart health technology firm, has officially launched the EarSight Ultra X, a next-generation ear care device featuring real-time 5G connectivity, 4K endoscopy imaging, and multiple safety enhancements tailored for family use. Positioned as a proactive wellness solution rather than a regulated diagnostic tool, the product aims to bring clinical-grade visualization into home environments, targeting common otologic concerns such as cerumen management, earbud-related hygiene issues, and trapped water in the ear canal.
The launch places Bebird at the center of a fast-emerging category within the consumer health sector, where diagnostic-quality imaging is migrating from clinical settings into the hands of everyday users. The broader implications for safety, adoption, and regulatory evolution could make EarSight Ultra X an important case study in the convergence of personal care devices and med-tech functionality.
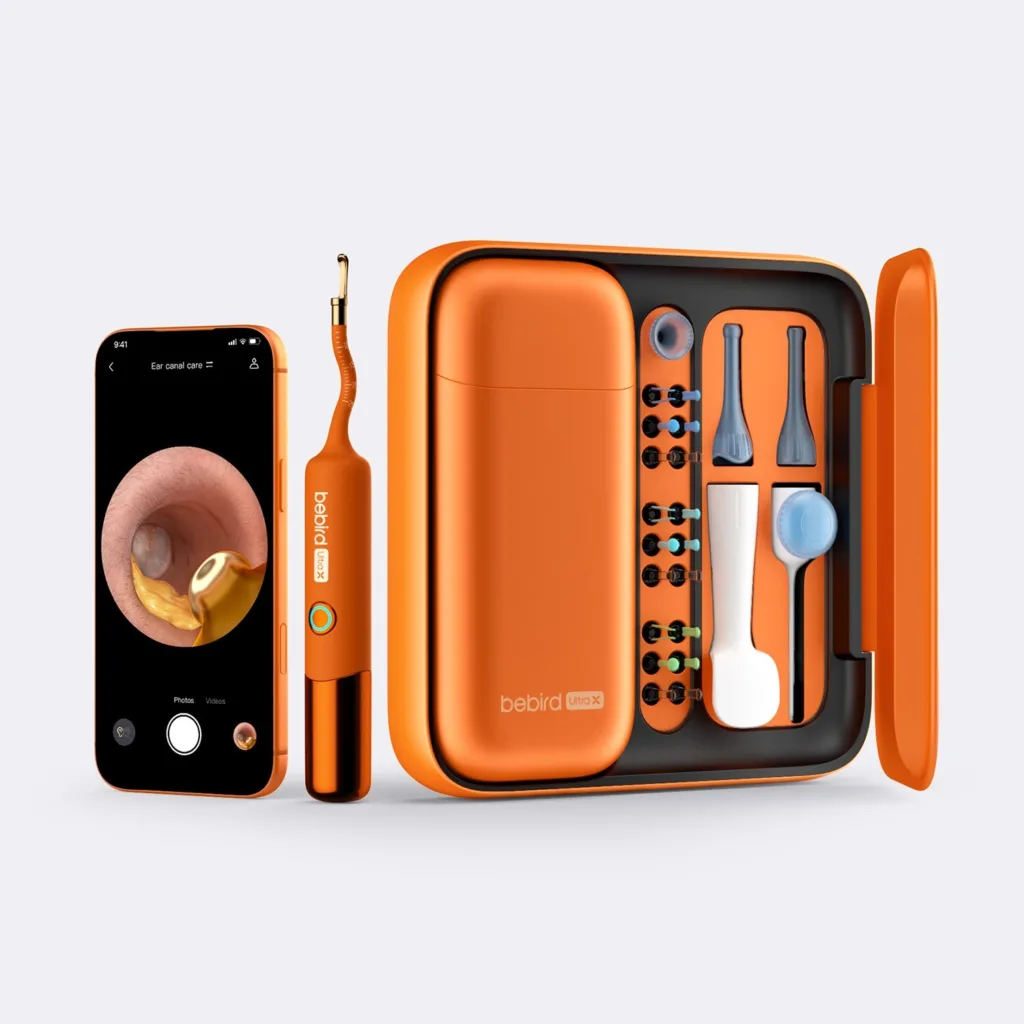
What this reveals about the evolving line between wellness tech and clinical-grade imaging
The defining feature of Bebird’s EarSight Ultra X is its use of a 5G-enabled chipset that allows high-definition, zero-latency video streaming from an embedded 4P endoscopic camera. The ability to capture and transmit real-time 4K imagery from within the ear canal not only improves visualization for parents performing ear cleaning at home but also raises expectations around what consumer ear care devices should deliver.
This level of image quality has historically been the domain of clinical otoscopes and ENT-specific instruments. By embedding these capabilities into a consumer device, Bebird is challenging the traditional segmentation of wellness and medical technologies. In doing so, the company introduces a host of regulatory and operational complexities that are likely to come under increasing scrutiny as more such products enter the market.
Industry observers note that Bebird’s design approach reflects an understanding of this challenge. The device includes an adjustable depth stop collar to prevent over-insertion, a fixed 36°C temperature stabilizer to ensure user comfort, and a built-in multi-axis gyroscope that maintains spatial orientation during use. These design elements may not elevate the product to the level of a regulated diagnostic tool, but they collectively represent a significant advancement in consumer safety engineering.
Why the rise of smart otoscopes could disrupt early-stage screening in primary care
As with many digital health innovations, the underlying question is not whether the technology works, but whether it changes behavior. Bebird’s EarSight Ultra X invites parents to take a more hands-on role in otologic screening and hygiene for their children, transforming ear cleaning from an intuitive or reactive routine into a visually guided, controlled procedure.
In theory, this can help reduce unnecessary physician visits for non-urgent concerns like wax buildup, while potentially flagging more serious issues earlier. However, there is a flip side. Without clinical training, users may misinterpret visuals, leading to either overconfidence or unnecessary anxiety. This risk is especially salient in pediatric care, where symptoms like mild inflammation, fluid retention, or anatomical variation can present subtly and require experienced interpretation.
Clinicians tracking the field believe this could become a friction point between empowered consumers and overstretched practitioners. On one hand, smart otoscopes like EarSight Ultra X could support remote care and pre-visit screening if integrated with telehealth platforms. On the other, they may contribute to diagnostic noise if users begin relying solely on subjective, visual impressions.
Bebird has thus far chosen not to position the device for clinical integration, and there is no mention of artificial intelligence or diagnostic overlays within the current product framework. However, the company’s choice to emphasize professional-grade clarity and real-time image control may lay the foundation for future integration with AI-based assistive tools or physician-reviewed teleconsultation platforms.
What this changes for consumer medical device manufacturers entering regulated gray zones
The EarSight Ultra X highlights a critical inflection point for consumer medical device manufacturers. As technical capability outpaces regulatory clarity, the question of whether a device qualifies as a wellness tool or a clinical instrument becomes less about hardware and more about intention, marketing, and secondary use.
In Bebird’s case, the device includes multiple risk-reduction design elements such as individual user kits to prevent cross-contamination, a sealed ear-drop dispenser attachment, and stabilization controls designed to reduce injury risk during use. However, none of these features trigger regulatory classification as a medical device in most jurisdictions, as long as the device is not explicitly marketed for diagnosis or treatment.
This ambiguity creates both opportunity and risk. For companies like Bebird, the ability to offer quasi-clinical features without a full regulatory burden may accelerate product cycles and broaden market access. However, the longer-term risk is that regulators may choose to reinterpret such devices retroactively, especially if adverse events or misleading use cases begin to surface.
From a competitive standpoint, Bebird’s move may also accelerate investment into adjacent categories such as smart dermatology scopes, at-home dental imaging, or gastrointestinal cameras designed for consumer use. The challenge for manufacturers will be to navigate safety, user training, and regulatory gray zones while still delivering consumer-level price points and usability.
What could limit uptake despite the technological differentiation
Despite its technical appeal, the EarSight Ultra X faces real barriers to mainstream adoption. Price sensitivity may be the most immediate challenge. While Bebird has not disclosed pricing in its official announcement, the inclusion of a high-end camera module, 5G chip, and precision safety mechanics suggests a retail tier above typical consumer wellness accessories.
For many households, especially in emerging markets or low-income settings, ear care is not considered a high-frequency need warranting capital outlay. Unless bundled with other health benefits or offered under promotional pricing, the device may remain a niche product for tech-forward or health-conscious parents.
Another constraint is interpretive confidence. Seeing inside the ear canal in 4K is only useful if the viewer can make sense of what they see. Without training or AI-driven interpretation, the risk of overconfidence or false reassurance could discourage repeat usage or even lead to harm. The need for better user education, clinical guardrails, or possible app-based support tools will become more pressing if Bebird aims to scale adoption beyond early tech adopters.
Finally, data privacy could become a limiting factor in future iterations. While current models appear to operate entirely on-device, any move toward app-based storage, cloud synchronization, or AI analysis will trigger scrutiny over personal health data handling. Given the sensitivity of video feeds involving children’s health, companies operating in this space will need to align with evolving global data protection standards, including HIPAA, GDPR, and regional equivalents.
What clinicians and regulators may monitor in the next iteration
The success or failure of Bebird’s EarSight Ultra X may ultimately rest on its ability to influence user behavior without crossing into regulated territory. The device is not a replacement for clinical otoscopy, nor does it currently offer diagnostic support. But by introducing visual clarity, safety, and interactivity into a wellness routine, it repositions consumer ear care from guesswork to guided practice.
Regulators may begin monitoring how users employ such devices in real life, particularly whether usage leads to delays in professional diagnosis, inappropriate self-treatment, or the emergence of unsanctioned secondary markets in app-based diagnosis. If Bebird or other manufacturers introduce AI-enabled interpretation or data-sharing features in future updates, the regulatory status of such devices could shift quickly.
Clinicians, particularly those in pediatrics and ENT, will also watch whether such tools become integrated into patient interactions. From showing visuals during virtual consults to enabling asynchronous pre-screening, the pathway to partial clinical integration is already visible. What remains unclear is whether manufacturers will lean into that direction or remain firmly within the wellness lane.
In the meantime, EarSight Ultra X has carved out a compelling niche: a high-spec, safety-oriented ear care platform for modern families. Whether it evolves into something more powerful or simply sets a higher bar for consumer-grade health tools, its impact on the personal diagnostics landscape is likely to be lasting.