Can Scribe Therapeutics make one-dose LDL-C lowering a practical CRISPR frontier?
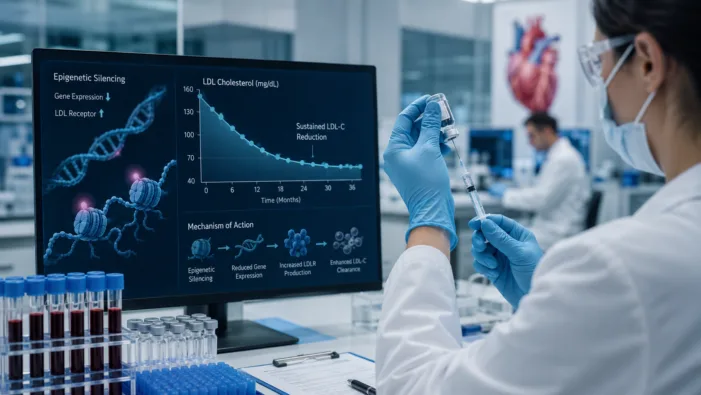
Find out how Scribe Therapeutics’ STX-1150 could reshape long-acting LDL-C lowering through CRISPR-based epigenetic silencing.

Find out how Scribe Therapeutics’ STX-1150 could reshape long-acting LDL-C lowering through CRISPR-based epigenetic silencing.

CRISPR can lower cholesterol. The harder test is whether Scribe can make durability, precision, and scalability work in common disease.

Viral vectors built gene therapy’s first era. Safety, redosing and manufacturing pressure are pushing non-viral platforms into the spotlight.

Origin Genomics launches in New York to advance germline gene correction and MRT under IRB oversight. Analyse what it means for US gene editing regulation.

Illumina, Inc. has launched the Billion Cell Atlas, a large-scale genetic perturbation dataset designed to serve as foundational infrastructure for artificial intelligence-enabled drug discovery. Announced alongside strategic collaborations with AstraZeneca, Merck, and Eli Lilly and Company, the platform leverages CRISPR-based genome editing and single-cell RNA sequencing across more than 200 disease-relevant cell lines. This marks […]

Can Aurora Therapeutics make CRISPR work at scale? Find out how its platform model for rare disease editing is testing biotech’s next big leap.

GenEditBio Limited, a Hong Kong–headquartered clinical-stage biotechnology firm focused on in vivo genome-editing therapies, announced that the United States Food and Drug Administration has cleared its Investigational New Drug application for GEB‑101, a CRISPR-based candidate for TGFBI corneal dystrophy. This regulatory milestone authorizes the launch of the Phase 1/2 CLARITY trial in the United States, […]