Can low-dose radiotherapy reshape osteoarthritis care after Siemens Healthineers’ FDA clearance
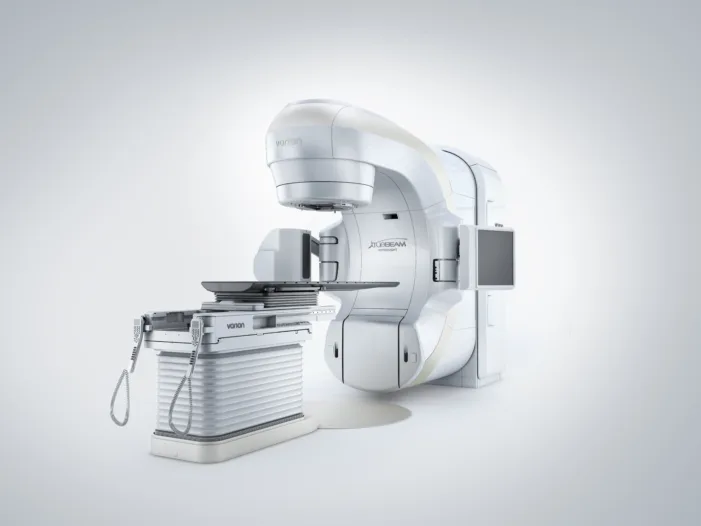
Siemens Healthineers expands TrueBeam into osteoarthritis care with FDA clearance. Find out how radiotherapy could reshape chronic pain treatment.

Siemens Healthineers expands TrueBeam into osteoarthritis care with FDA clearance. Find out how radiotherapy could reshape chronic pain treatment.

FDA clears expanded capabilities for Bayer MRXperion MRI injector supporting scanners up to 7T. Discover what this means for imaging workflows and radiology systems.
GE HealthCare’s FDA-cleared SIGNA MRI systems signal a shift toward autonomous, helium-free imaging. Explore what this means for MRI operations.

Bimini Health Tech secures FDA clearance for Puregraft SYNC. Explore what it changes for fat grafting workflows, adoption economics, and device strategy.