Why Galderma’s acne gel approval could quietly reset the over the counter dermatology market

Acne care is moving from prescription desks to retail shelves. Galderma’s Differin Epiduo switch tests how far OTC dermatology can go.

Acne care is moving from prescription desks to retail shelves. Galderma’s Differin Epiduo switch tests how far OTC dermatology can go.

Galderma has launched Regenerating Skin Nectar with TriHex+. Read why this matters for peri-procedural skincare, clinics, and regenerative aesthetics.

Read how Galderma’s new interim data could influence aesthetic treatment planning in menopause and GLP-1-related volume loss.

Galderma’s ALASTIN Signature Practices aim to embed skincare into aesthetic workflows. Read what this means for clinics, competition, and growth.
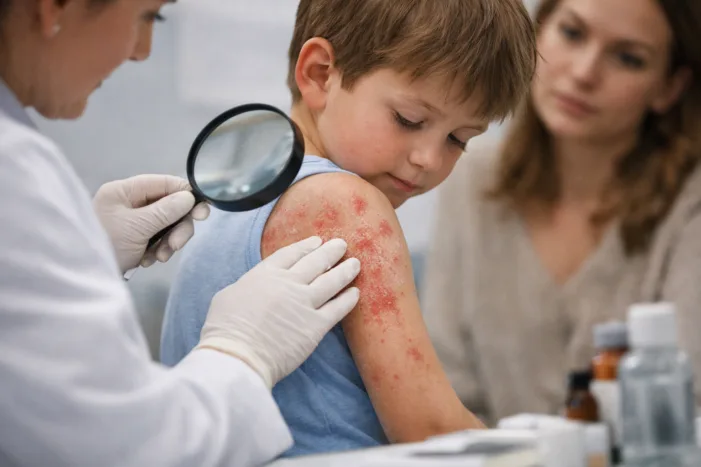
Galderma’s nemolizumab phase II data in children aged 2-11 show 52-week itch and lesion relief. Read the analysis.

FDA approval of Restylane Contour for temple hollowing signals a shift in facial aesthetics. Discover what this means for the industry and future treatments.

Galderma launches a two-year Sculptra initiative tracking real-world outcomes. Discover what this means for regenerative aesthetics and durability claims.

Explore how Galderma’s Wake Up To Restylane strategy and new IMCAS data could reshape aesthetic practice and hyaluronic acid filler adoption.