Why Miracell’s SMART M-CELL FDA clearance could reshape the PRP and BMAC device market

Miracell has won FDA 510(k) clearance for SMART M-CELL. Read what it changes for PRP, BMAC, adoption, and regenerative medicine competition.

Miracell has won FDA 510(k) clearance for SMART M-CELL. Read what it changes for PRP, BMAC, adoption, and regenerative medicine competition.
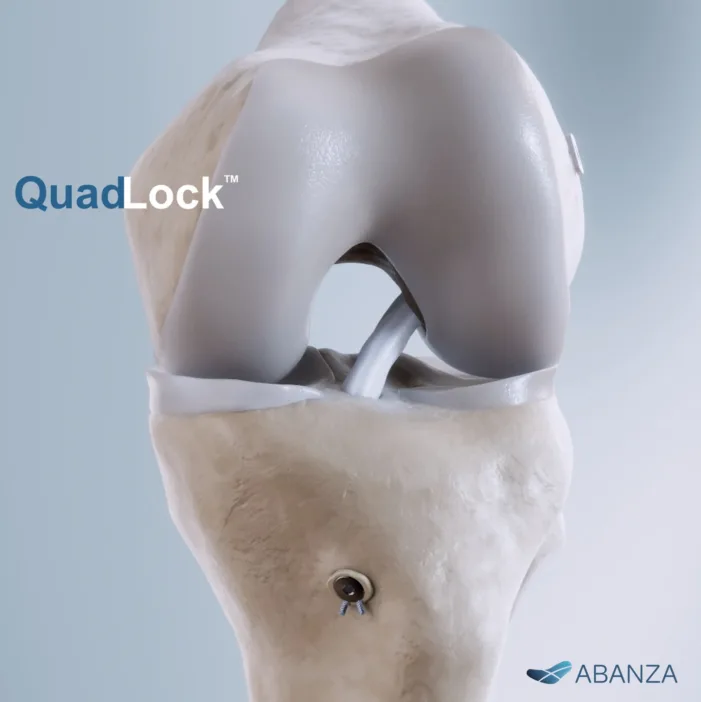
Abanza Inc. has received U.S. Food and Drug Administration (FDA) 510(k) clearance for QuadLock, its novel knotless fixation system for anterior cruciate ligament (ACL) reconstruction. The system, which features a bidirectional tension-adjustable design, reportedly offers over a 500% reduction in cyclic displacement compared to conventional fixation methods such as cortical buttons and interference screws, targeting […]

FDA clears PTOA risk reduction claim for Miach Orthopaedics’ BEAR Implant. Find out what this means for ACL treatment, patients, and future adoption.