Obodence at WCO 2026: Samsung Bioepis sharpens the clinical case for SB16 adoption
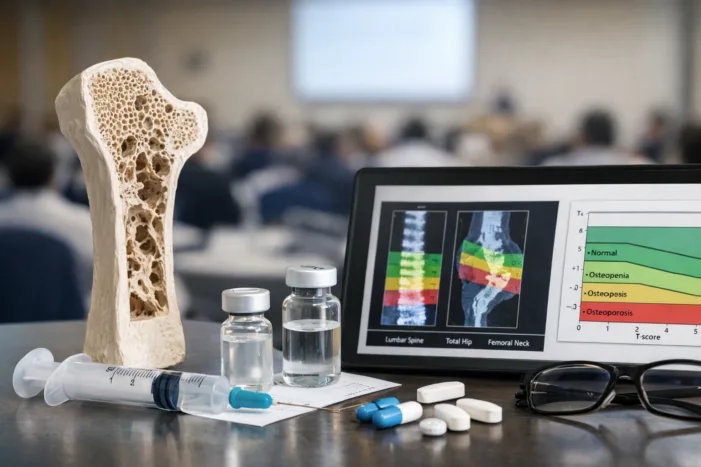
Samsung Bioepis presented new SB16 subgroup data at WCO 2026. Read what it means for Obodence adoption, biosimilar trust, and osteoporosis care.

Samsung Bioepis presented new SB16 subgroup data at WCO 2026. Read what it means for Obodence adoption, biosimilar trust, and osteoporosis care.
mAbxience and Amneal Pharmaceuticals have secured U.S. Food and Drug Administration (FDA) approval for two denosumab biosimilars, Boncresa and Oziltus, referencing Prolia and XGEVA respectively, clearing a long-anticipated regulatory hurdle in osteoporosis and oncology supportive care. The approvals position Amneal Pharmaceuticals as the exclusive U.S. commercial partner, with mAbxience responsible for development and manufacturing, marking […]

Sōlaria Biō has disclosed new mechanistic data showing that its synbiotic medical food, Bondia, reinforces gut barrier integrity, reduces systemic inflammation, and suppresses osteoclast activity—offering biological insight into the 85 percent improvement in bone density reported in a prior large-scale clinical trial. These results support Bondia’s positioning as a novel intervention for osteopenia and age-related […]