Samsung Bioepis Co., Ltd. used WCO-IOF-ESCEO 2026 to present subgroup data showing that SB16, marketed in Europe as Obodence, delivered efficacy consistent with reference denosumab across key osteoporosis patient populations in a Phase 3 study of postmenopausal women. The update matters because Obodence already holds European Union authorization and has been commercially launched in Europe, so the new analysis is less about winning approval and more about reducing the practical hesitation that still shadows biosimilar prescribing in chronic bone health.
Why subgroup consistency matters more than it first appears in osteoporosis biosimilar adoption
For osteoporosis specialists, subgroup consistency is not a decorative secondary analysis. It goes straight to the real-world question of whether a biosimilar can be used with confidence in the messy clinical population that appears in fracture clinics, endocrine practices, and aging-medicine settings. A biosimilar can meet analytical and primary clinical comparability thresholds, yet still face softer resistance if prescribers worry that treatment effect may drift in older patients, in women with lower body mass index, in prior bisphosphonate users, or in patients carrying baseline vertebral fracture burden. That is why Samsung Bioepis’ emphasis on age, body mass index, smoking history, prior oral bisphosphonate use, and vertebral fracture status is strategically well chosen. It addresses the exact places where clinician caution tends to hide even after regulators have already accepted the totality of evidence. The limitation, however, is that subgroup work generally reassures rather than transforms. It rarely creates new differentiation, and because subgroup analyses can be underpowered for fine-grained conclusions, it is best understood as confidence-building evidence rather than a new clinical breakthrough.
How the Phase 3 design supports biosimilarity, while still leaving familiar adoption questions
The underlying Phase 3 trial enrolled 456 postmenopausal women with osteoporosis in a randomized, double-blind, multicenter design comparing SB16 with reference denosumab. Efficacy was assessed through percentage change in bone mineral density at the lumbar spine, total hip, and femoral neck at month 12, with comparative analyses performed using analysis of covariance. In biosimilar development, that framework is exactly what regulators expect. The goal is not to re-prove de novo clinical superiority or long-term fracture reduction in a giant outcomes study, but to demonstrate no clinically meaningful difference in pharmacology, efficacy, safety, and immunogenicity versus the reference product in a sensitive population. That gives the dataset regulatory strength. The unresolved issue is that osteoporosis prescribers do not always think in pure biosimilarity logic. Many still care more about fracture outcomes, persistence, switching behavior, and post-marketing comfort than about the elegant architecture of a comparative equivalence package. So while the trial design is fit for purpose, commercial uptake will still depend on how convincingly Samsung Bioepis and health systems translate biosimilar science into day-to-day clinical trust.
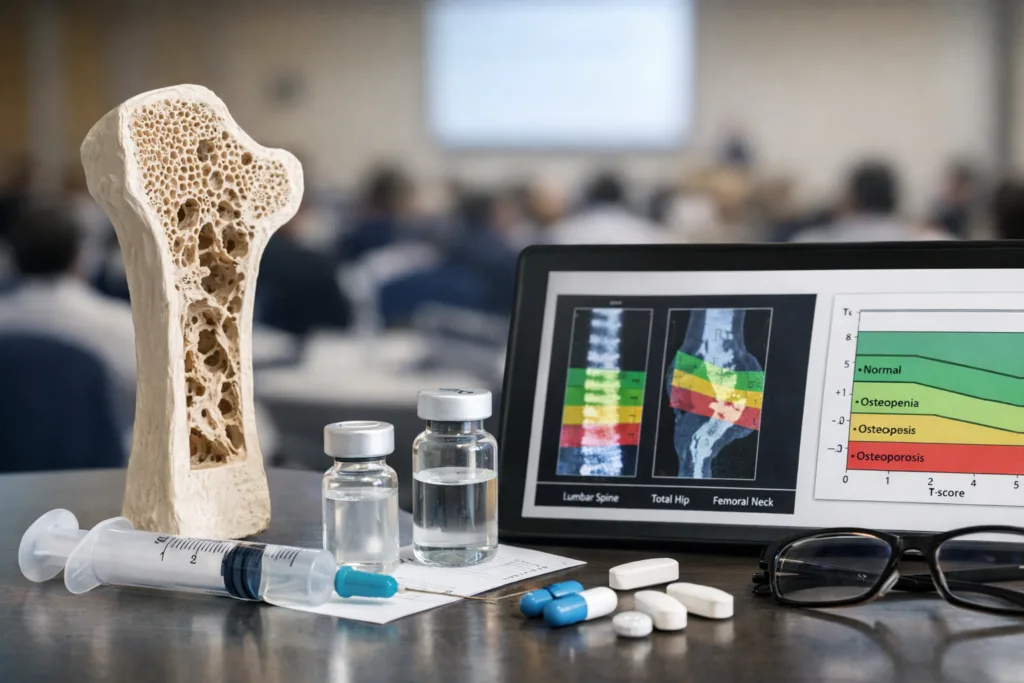
Why this poster is really about physician confidence, not regulatory validation anymore
Obodence received European Union marketing authorization on February 12, 2025, after the European Medicines Agency reviewed it as a biosimilar to Prolia. Samsung Bioepis then launched Obodence in Europe in December 2025, which means the product has already crossed the highest formal regulatory hurdle and entered the commercial arena. Against that backdrop, the Prague poster functions as a post-approval confidence signal. It helps the company reinforce that the biosimilarity case is durable when viewed through subgroup lenses that matter to prescribers. That is commercially meaningful in osteoporosis, where treatment decisions can be sticky and switching patients from a familiar originator product is often slower than tender logic alone would suggest. The risk is that post-approval scientific communication can only do so much if payer policy, local procurement structures, or physician habits remain conservative. Biosimilar uptake is never driven by clinical comparability data alone. It also depends on contracting, education, distribution readiness, and the willingness of health systems to operationalize substitution or switching pathways.
What this reveals about the next battleground in denosumab biosimilars across Europe
The denosumab category is becoming more crowded, and that changes the stakes. Obodence is not entering a pristine market where being first with acceptable data is enough. European regulators have also assessed other denosumab biosimilar dossiers, underlining that the real contest is shifting from approval to positioning. In that environment, subgroup consistency becomes part of a larger competitive narrative. It helps Samsung Bioepis argue that its package is not merely adequate but robust enough to support broad confidence across routine osteoporosis populations. That message matters to hospital pharmacists, national tender bodies, and specialists weighing whether one biosimilar has a cleaner evidence story than another. Still, there is a ceiling to how much scientific nuance can differentiate products that regulators already consider highly similar to the same reference medicine. Once multiple denosumab biosimilars are in play, price, supply assurance, device familiarity, and commercial execution may matter more than marginal differences in how persuasively each sponsor presents subgroup reassurance.
How totality-of-evidence arguments keep driving biosimilars beyond headline efficacy data
Samsung Bioepis framed the poster as reinforcement for the totality-of-evidence supporting SB16 biosimilarity, and that phrase is doing important work. Biosimilar regulation does not rest on a single efficacy endpoint or one headline clinical result. It rests on a layered package spanning analytical similarity, functional characterization, pharmacokinetics, pharmacodynamics where relevant, immunogenicity, safety, and confirmatory clinical comparison in a sensitive setting. The subgroup analysis fits neatly into that architecture because it adds another lens through which comparability can be viewed. For industry observers, that is the correct scientific framing. For clinicians, though, totality-of-evidence can still feel abstract compared with practical concerns such as injection-device handling, continuity of supply, reimbursement criteria, and how to manage therapy interruption. Denosumab carries clinically important discontinuation considerations in osteoporosis care, so adoption depends not only on proving biosimilarity at baseline, but on ensuring confidence in treatment continuity and system-level execution over time.
Why the commercial significance extends beyond osteoporosis into Samsung Bioepis’ portfolio strategy
Obodence also matters because it extends Samsung Bioepis into endocrinology, adding a new therapeutic pillar beyond the company’s more established biosimilar footprint. Both the European and United States approvals marked the denosumab programs as the company’s first endocrinology biosimilars, and that broadens Samsung Bioepis’ strategic identity from a biosimilar manufacturer with depth in select categories into a company pursuing wider therapeutic range and more direct commercialization. That portfolio logic matters in Europe, where the company has been expanding its direct commercial presence. A successful denosumab rollout would not just generate revenue from one molecule. It would strengthen Samsung Bioepis’ case that it can manage increasingly complex launches across therapeutic areas where physician education and long-term treatment confidence are central. The remaining question is whether expansion breadth can be matched by enough commercial focus. Entering more categories is attractive, but each one comes with its own adoption psychology, payer dynamics, and competitive intensity. Osteoporosis is not immunology by another name, and execution shortcuts tend to get exposed quickly in chronic care markets.
What clinicians and market watchers are likely to watch next after this subgroup update
The immediate takeaway is that Samsung Bioepis has added another layer of reassurance to an already approved and launched biosimilar. The next phase of scrutiny, however, will be less about abstract comparability and more about market behavior. Clinicians will watch whether confidence in switching and initiation broadens in daily practice. Procurement teams will watch price competitiveness and supply consistency. Regulators will continue to expect pharmacovigilance discipline rather than celebratory interpretation of subgroup posters. Competitors will watch whether Samsung Bioepis can convert scientific completeness into formulary wins before the denosumab biosimilar field becomes too crowded for evidence packages alone to stand out. In other words, the Prague data may not change the science of biosimilarity, but it could still matter if it helps remove one more layer of friction between approval and routine use. That is usually where biosimilar stories are actually won or lost.