Why DePuy Synthes’ Gemtrack deal could reshape navigation in hip, knee and shoulder surgery

DePuy Synthes wants to remove a robotics bottleneck. Its Gemtrack deal could change joint surgery navigation if regulators agree.

DePuy Synthes wants to remove a robotics bottleneck. Its Gemtrack deal could change joint surgery navigation if regulators agree.

Pixee Medical’s Knee+ NexSight receives CE mark. Analyse what AR guidance offers over robotics in total knee arthroplasty and what questions remain.
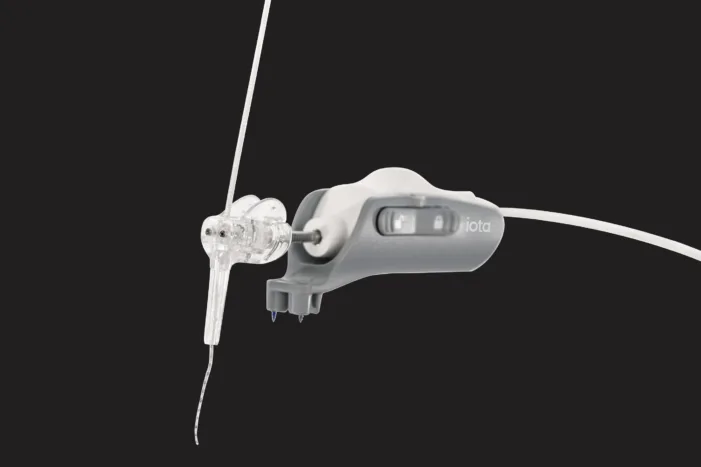
iotaMotion, Inc. has received U.S. Food and Drug Administration 510(k) clearance to expand the approved use of its iotaSOFT Robotic-Assisted Cochlear Implant Insertion System for children as young as four years old. This regulatory milestone makes the iotaSOFT system the first robotic-assisted cochlear insertion platform authorized for use in school-aged pediatric patients, expanding access to […]