iotaMotion, Inc. has received U.S. Food and Drug Administration 510(k) clearance to expand the approved use of its iotaSOFT Robotic-Assisted Cochlear Implant Insertion System for children as young as four years old. This regulatory milestone makes the iotaSOFT system the first robotic-assisted cochlear insertion platform authorized for use in school-aged pediatric patients, expanding access to precision-guided cochlear surgery.
The FDA’s decision signals a shift in clinical expectations for cochlear implant surgery precision
The FDA’s clearance is more than a label expansion. It represents a potential inflection point in how cochlear implant surgery is performed and evaluated, especially in pediatric populations where the stakes of hearing preservation and neurodevelopmental outcomes are particularly high. The iotaSOFT system enables robotic control during one of the most delicate steps of cochlear implantation: the insertion of the electrode array into the cochlea.
This stage is often vulnerable to human variability, with potential trauma to the cochlear structures impacting both immediate auditory performance and long-term compatibility with emerging regenerative therapies. By extending regulatory approval to younger patients, the FDA has effectively endorsed robotic consistency as a viable strategy to address these risks, opening the door for broader pediatric application of surgical robotics in otology.
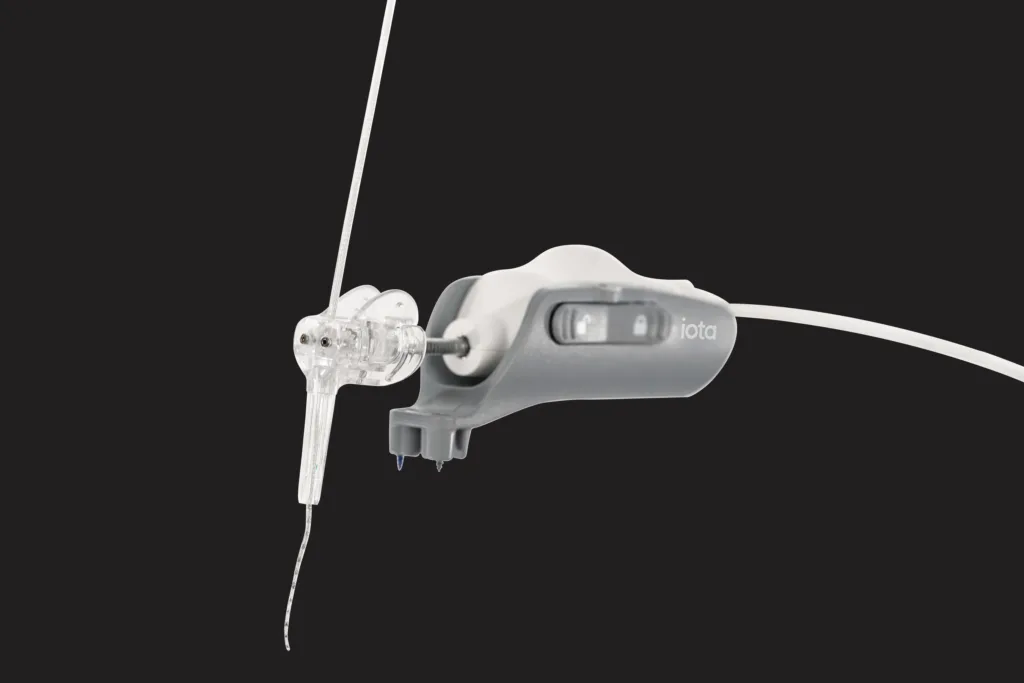
Why pediatric approval fundamentally raises the bar for cochlear implantation protocols
Pediatric hearing restoration is a clinical domain driven by early intervention and neural plasticity. The earlier a child receives access to sound, the greater the potential for language development and cognitive alignment. However, traditional cochlear implantation methods have always involved trade-offs between access and preservation, particularly for children with residual hearing or those expected to benefit from future therapeutic advances.
In this context, the iotaSOFT platform’s ability to standardize electrode insertion through controlled velocity and depth offers a surgical safeguard. Industry analysts suggest that robotic platforms like iotaSOFT could eventually become the procedural gold standard, particularly as healthcare systems and payers increasingly prioritize outcomes that extend beyond initial activation to include hearing preservation, therapy compatibility, and long-term auditory function.
Early adoption by Cincinnati Children’s validates the system’s clinical traction
The announcement that Cincinnati Children’s Hospital is the first pediatric center to adopt the iotaSOFT system following the expanded clearance is significant. As one of the top-ranked pediatric hospitals in the United States and a consistent early adopter of high-impact medical technologies, Cincinnati Children’s sets a precedent that may influence other major pediatric ENT programs.
Institutional adoption is often a bellwether for systemic change in surgical disciplines. Cincinnati’s move suggests not only internal confidence in the technology’s risk-benefit profile but also a growing alignment between robotics and standard-of-care expectations in pediatric auditory surgery. The hospital joins more than 35 cochlear implant centers in the United States that have already integrated iotaSOFT into their surgical programs, a roster that includes nearly half of all U.S. neurotology fellowship training institutions.
Clinical data points to meaningful gains in hearing preservation but more validation is needed
A 2025 cohort study published in The Laryngoscope offers preliminary evidence in support of robotic-assisted cochlear insertion. According to the authors, 85 percent of patients who underwent robotic-guided insertion preserved residual hearing at one year, compared to 71 percent in the manual insertion group. While these figures are encouraging, they are not yet definitive.
Clinicians following the space emphasize that real-world performance across diverse pediatric anatomies remains to be fully characterized. Variability in cochlear size, ossification patterns, and patient-specific surgical challenges could influence how well robotic precision translates into consistent pediatric outcomes. Additionally, the technology does not yet offer real-time feedback mechanisms for insertion pressure or tissue resistance, which are areas where future iterations of robotic platforms could provide further clinical control.
The reimbursement and workflow puzzle may slow broader adoption despite clinical momentum
One of the key friction points for hospitals considering robotic surgical systems is not clinical validation but reimbursement feasibility. At present, robotic cochlear implantation does not carry differentiated procedural codes in most payer systems. As a result, institutions must justify the capital investment in iotaSOFT based on internal performance metrics, training advantages, or anticipated reductions in complications.
Moreover, while robotic insertion may improve precision, it can also extend surgery time, especially in centers without dedicated robotic ENT teams. Operating room efficiency remains a core KPI for many hospitals, and unless improved outcomes lead to lower revision rates, better hearing thresholds, or expanded patient access, the financial case for robotics may remain confined to top-tier academic or specialty programs.
Nonetheless, as payer models shift toward bundled payments and long-term quality metrics, robotics may gain traction as a risk-reduction tool in complex auditory procedures, particularly for children.
Regulatory clarity sets the stage for future integration with regenerative and gene-based therapies
Beyond immediate surgical benefits, one of the most consequential aspects of iotaSOFT’s expansion into pediatrics is its alignment with the needs of future therapeutic modalities. Several gene therapies and regenerative interventions currently in early-stage development depend on atraumatic access to the cochlea. Whether for delivery of viral vectors, stem cells, or molecular therapies, an intact cochlear microenvironment is often a precondition.
In this context, platforms like iotaSOFT may become surgical enablers of biologic therapies that are still years from commercial readiness. This convergence between mechanical precision and biologic delivery has already been seen in other fields such as ophthalmology and neurosurgery. In pediatric cochlear implant candidates, who may be ideal patients for future re-intervention or augmentation therapies, the value of preserving cochlear integrity early in life cannot be overstated.
Stakeholders tracking this field suggest that robotic cochlear insertion could eventually evolve from an optimization strategy to a foundational requirement in cases where structural preservation is essential for therapeutic eligibility.
What this means for the future of robotic systems in ENT surgery and pediatric innovation
The expanded clearance for iotaMotion’s iotaSOFT platform offers a glimpse into a broader transformation underway in pediatric surgical care. As robotics continue to move from orthopedics and cardiology into more delicate, microstructural domains like otolaryngology, the question is no longer whether robotic precision is feasible, but whether it is necessary.
For medical device manufacturers, the pediatric use case opens a new commercial frontier. For hospitals, it presents an opportunity to lead with clinical innovation in an area where hearing loss remains under-treated and where precision can have a lifelong impact. For regulators, the decision represents a cautious but meaningful signal that the benefit-risk calculus for surgical robotics is evolving—even in vulnerable patient populations.
As fewer than 5 percent of the estimated 60 million cochlear implant candidates worldwide receive implants today, largely due to variability in outcomes and access concerns, the emergence of standardized robotic solutions could help address some of these systemic gaps.
Whether robotic insertion becomes the new norm or remains a specialized adjunct will depend on post-clearance performance data, real-world adoption metrics, and evolving stakeholder consensus on what constitutes procedural excellence in auditory restoration.