Can non-viral gene therapies solve the safety problem holding back genetic medicine?

Viral vectors built gene therapy’s first era. Safety, redosing and manufacturing pressure are pushing non-viral platforms into the spotlight.

Viral vectors built gene therapy’s first era. Safety, redosing and manufacturing pressure are pushing non-viral platforms into the spotlight.

Read why Sarepta Therapeutics’ first siRNA data in FSHD1 and DM1 could matter for rare disease drug development and investor sentiment.
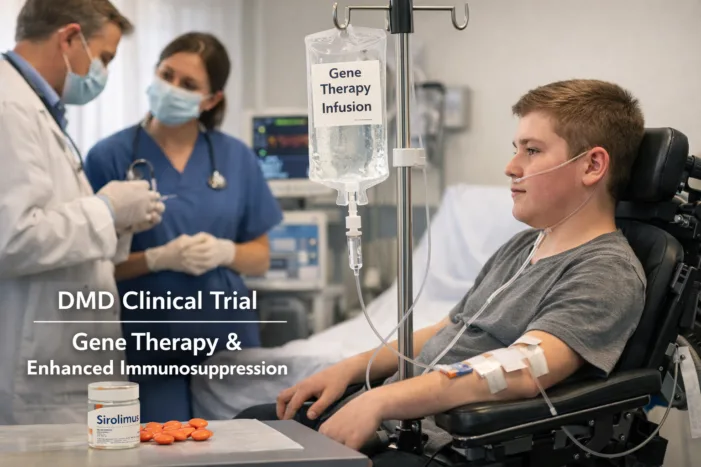
Sarepta’s ELEVIDYS Cohort 8 targets non-ambulatory Duchenne patients with a sirolimus regimen. What the trial design reveals about the path to label restoration.

Sarepta’s gene therapy ELEVIDYS shows 3-year durability in Duchenne muscular dystrophy. Find out what it means for patients, payers, and global rollout.

Sarepta’s EMBARK 3-year readout could clarify ELEVIDYS’ long-term role in Duchenne care. Find out what’s at stake in this pivotal gene therapy trial.