GT Medical Technologies has announced the first patient enrollments in its BRIDGES trial, a randomized clinical study assessing the use of GammaTile therapy in newly diagnosed glioblastoma. The trial introduces GammaTile at the time of initial tumor resection, marking a notable shift from the conventional glioblastoma radiation timeline, which typically delays radiation until several weeks post-surgery. By embedding the radiotherapy device intraoperatively, GT Medical Technologies aims to deliver localized treatment immediately following resection, potentially addressing the rapid regrowth risks associated with the standard of care.
What the BRIDGES trial reveals about rethinking radiation timing in glioblastoma
The central premise of the BRIDGES trial is to test whether radiation delivered at the time of surgery can improve survival outcomes in glioblastoma, a cancer defined by its aggressiveness and poor prognosis. The traditional model of care has long featured a gap of several weeks between surgical resection and the onset of radiotherapy. While intended to allow patient recovery and treatment planning, this interval creates an opportunity for residual tumor cells to begin repopulating, especially in a disease where recurrence is more the norm than the exception.
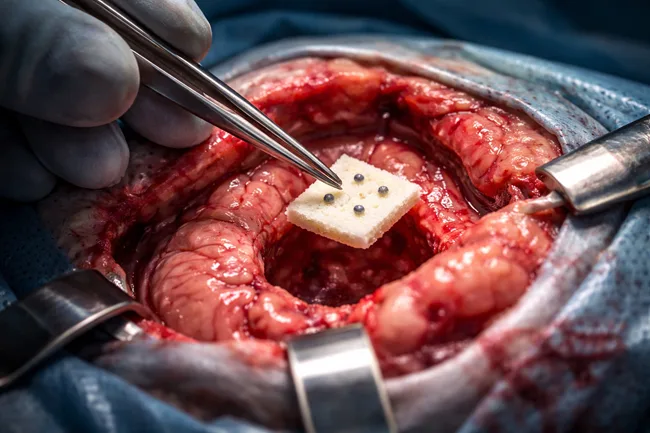
GammaTile, an FDA-cleared bioabsorbable collagen matrix embedded with cesium-131 radiation seeds, is designed to be implanted during surgery. It delivers a sustained local radiation dose over several weeks, bypassing the typical lag between excision and external beam radiation therapy. Clinical observers believe this timing could be particularly impactful in glioblastoma, where tumor cells are known to rapidly invade surrounding brain tissue even before visible regrowth.
The BRIDGES trial seeks to provide the evidence needed to support a revision of treatment standards. While the concept of intraoperative radiation is not new, its integration into routine glioblastoma workflows has remained limited, partly due to logistical challenges and limited device approvals. GammaTile may be the first to demonstrate operational feasibility and scalable deployment within existing surgical and oncology settings, especially if BRIDGES meets its clinical endpoints.
Why GammaTile is positioned differently from prior intraoperative radiation approaches
GammaTile’s design addresses several historical limitations of intraoperative radiation in neurosurgery. Unlike approaches such as intraoperative electron radiation therapy or shielded devices requiring specialized equipment, GammaTile requires no external machinery. It consists of collagen tiles preloaded with radiation sources, which are placed along the tumor cavity margins at the end of resection. The embedded radiation is released over a fixed time window, targeting residual cells precisely when tumor burden is lowest.
This avoids the need for patients to return weeks later for radiation planning and delivery, a process that adds delay and demands logistical coordination between neurosurgery and radiation oncology teams. In practice, GammaTile transforms the resection itself into the start of radiation therapy.
GammaTile has already received FDA 510(k) clearance for use in recurrent glioblastoma and brain metastases. The BRIDGES trial is the first to evaluate it in the frontline setting for newly diagnosed GBM. Clinicians and device specialists tracking the field suggest this represents the therapy’s most ambitious application yet. Early adoption has occurred primarily in academic medical centers, and expanding to broader hospital systems will require stronger evidence.
How BRIDGES builds on GT Medical Technologies’ prior clinical momentum
The BRIDGES trial follows GT Medical Technologies’ completion of the ROADS study in patients with brain metastases. While results from that trial have yet to be publicly released, the company’s decision to proceed with BRIDGES signals internal confidence in GammaTile’s ability to impact outcomes when introduced earlier in the treatment sequence.
With the glioblastoma standard of care remaining relatively static for more than a decade, there is both clinical urgency and commercial opportunity for approaches that can extend survival without adding procedural complexity or post-operative burden. The timing of GammaTile implantation—during a surgical intervention already indicated for diagnosis or cytoreduction—makes it attractive to surgeons and health systems alike, particularly if reimbursement pathways can be aligned.
That said, GammaTile’s clinical profile must stand up to comparison with both standard radiation and emerging alternatives. The most recent addition to the glioblastoma device landscape, Tumor Treating Fields by Novocure, has seen limited uptake outside major centers despite FDA approval and modest overall survival gains. Observers suggest that the path forward for GammaTile may rely more on integration and workflow simplicity than on standalone efficacy.
What clinicians and regulators will watch as BRIDGES unfolds
A critical area of interest will be the BRIDGES trial’s primary endpoints and stratification across molecular glioblastoma subtypes. Details of the full study design have not yet been made public, but outcomes such as overall survival, progression-free survival, and rates of radiation necrosis will likely guide clinical interpretation and payer assessments.
The IDH-wild type and MGMT-unmethylated glioblastoma populations are known to respond poorly to conventional chemoradiation. If GammaTile shows a benefit in these high-risk subgroups, it could accelerate interest and adoption in academic and high-volume centers. Conversely, if benefits are confined to molecularly favorable cases, its market may be more limited.
Regulatory watchers note that the FDA has already classified GammaTile as a Class II device, allowing it to bypass lengthy premarket approval processes. However, broader reimbursement and pathway integration will likely hinge on BRIDGES producing peer-reviewed, statistically significant data across clinically relevant endpoints.
Moreover, real-world adoption will depend on demonstrating that GammaTile does not introduce new procedural risks such as radiation-induced tissue damage, cognitive deficits, or impaired wound healing. Early safety data and protocol adherence will be critical in these early trial sites.
Why glioblastoma remains a difficult proving ground for device innovation
Despite multiple experimental approaches, glioblastoma survival outcomes have remained largely unchanged for over two decades. Temozolomide chemotherapy, radiotherapy, and surgical resection form the current triad, with few successful additions to date. Numerous targeted therapies have failed to produce meaningful clinical benefit, and tumor heterogeneity continues to stymie drug development.
Device-based approaches have faced similar hurdles. Tumor Treating Fields faced slow uptake despite positive randomized data. Radiation strategies have long struggled to balance effective dosing with acceptable toxicity. The blood–brain barrier limits the efficacy of many systemically administered agents, giving localized solutions like GammaTile theoretical appeal.
That said, glioblastoma’s anatomical complexity, diffuse borders, and immunologically cold profile make it a challenging first indication. If BRIDGES succeeds, it would mark a rare instance of a device improving survival in primary brain tumors. But if results are inconclusive, it could reinforce skepticism about new technologies in this domain.
Clinicians have also flagged that surgical access remains a constraint. GammaTile is only usable when maximal safe resection is performed, which is not always possible depending on tumor location and patient comorbidities. This may inherently restrict the patient population eligible for treatment, even if the therapy proves effective.
What obstacles remain in commercializing GammaTile across broader practice settings
Even with a successful trial, GT Medical Technologies faces a number of systemic challenges. Reimbursement for intraoperative radiation varies by state, insurer, and hospital coding practices. Some centers may struggle to align OR and radiation planning workflows or justify upfront costs without compelling cost-offset data.
Manufacturing and supply chain scalability will also be tested if BRIDGES leads to broader demand. GammaTile requires sterile preparation and radiation safety handling protocols. Distribution across smaller hospitals or international markets may require partner support or regulatory harmonization beyond the United States.
Furthermore, integrating GammaTile into multimodal treatment regimens raises important clinical questions. Will it allow for de-escalation of external beam radiation? Can it be safely used alongside immunotherapies or targeted agents under investigation? Will it impact the timing or sequencing of standard chemotherapy regimens?
Until BRIDGES provides clarity on survival, tolerability, and recurrence dynamics, most of these remain speculative. Still, interest is likely to build as enrollment progresses and interim data become available.
Is this the start of a new paradigm in neuro-oncology?
The BRIDGES trial represents more than a single device study. It challenges the foundational assumption that radiation must be delayed after surgery, a convention that has shaped neuro-oncology practice for decades. If GammaTile can demonstrate that immediate intraoperative radiation improves outcomes without adding risk or burden, it may prompt a larger re-evaluation of how and when we treat glioblastoma.
For now, GT Medical Technologies has secured early traction with trial site activations at Westchester Medical Center and Brown University Health. But the true test will come when the data speaks. Until then, regulators, payers, and clinical stakeholders will be watching BRIDGES closely—not just for what it says about GammaTile, but for what it may signal about the future structure of brain cancer care.