What Galderma’s nemolizumab phase II data mean for the competitive biologic landscape in pediatric eczema
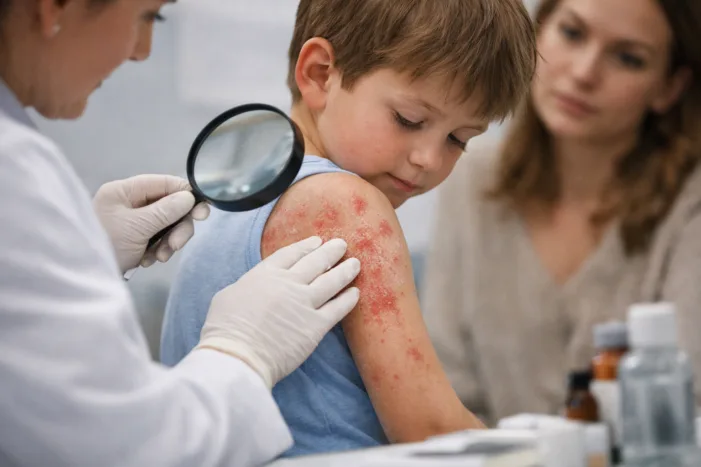
Galderma’s nemolizumab phase II data in children aged 2-11 show 52-week itch and lesion relief. Read the analysis.

Galderma’s nemolizumab phase II data in children aged 2-11 show 52-week itch and lesion relief. Read the analysis.

Kardigan’s tonlamarsen showed Phase 2 promise in hypertension, but can it win in acute severe hypertension next? Read the deeper analysis.

Anumana won FDA clearance for an ECG-AI pulmonary hypertension tool. Read what it changes for diagnosis, workflow adoption, and cardiovascular AI strategy.

Novo Nordisk’s Awiqli wins FDA approval as the first once-weekly basal insulin. Read what this could change for type 2 diabetes care.

Medtronic won FDA clearance for Stealth AXiS in cranial and ENT surgery. Read why this platform move could reshape surgical workflow competition.

FDA approves Agilent’s PD-L1 IHC 22C3 pharmDx for esophageal and GEJ carcinoma, marking the eighth companion diagnostic indication alongside pembrolizumab. Read the analysis.

Drug Farm won FDA orphan drug designation for DF-003 in ROSAH syndrome. Read what this rare disease milestone could change next.
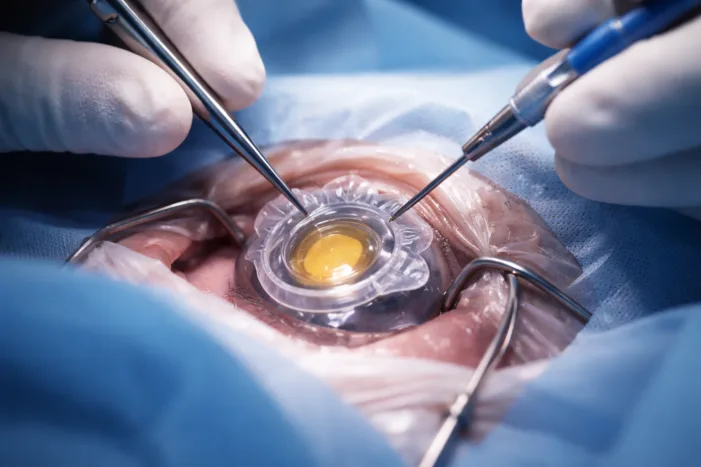
Long Bridge Medical’s LensOne has entered a pivotal FDA-cleared study. Read why this ophthalmic device trial could reshape secondary IOL fixation.

NEOK002 has cleared the FDA IND stage. Read why this bispecific ADC matters for solid tumors, clinical strategy, and the next phase of ADC competition.

AbbVie is repositioning Juvéderm for the “undetectable” aesthetics era. Read what this means for injectables, clinicians, and market competition.