Why Better Life Science’s Lyme testing push matters more for access than for diagnosis alone
Better Life Science is pushing at-home Lyme testing this spring. Read what Tick Bite Hero changes, where it fits, and what risks remain.

Better Life Science is pushing at-home Lyme testing this spring. Read what Tick Bite Hero changes, where it fits, and what risks remain.

Abbisko Therapeutics won EMA orphan drug designation for irpagratinib in liver cancer. Read what this changes for FGFR4-targeted therapy.

Tenpoint Therapeutics and PHIL launch YUVEZZI direct-to-patient access for presbyopia. Read what it means for affordability, uptake, and market adoption.

EarliPoint Health and Netsmart are expanding access to autism assessment technology. Read what this could change for providers, workflows, and adoption.

PharmaEssentia’s Puerto Rico facility could reshape BESREMi supply resilience and U.S. expansion. Read what this means for biologics strategy.

CTIS launches Trial SafetyXchange to help pharma firms meet the FDA E2B(R3) deadline. Read what it changes for safety reporting teams.

Aspect Biosystems has secured a $280 million Canada-backed push in regenerative medicine. Read what it means for cellular therapy scale-up.

OpenEvidence and Tandem are linking clinical AI with prior authorization automation. Read what this could change for prescribing, access, and workflow.
Can VR LIFE and H2 Global Group scale from European validation to global MedTech relevance? Read the deeper analysis now.
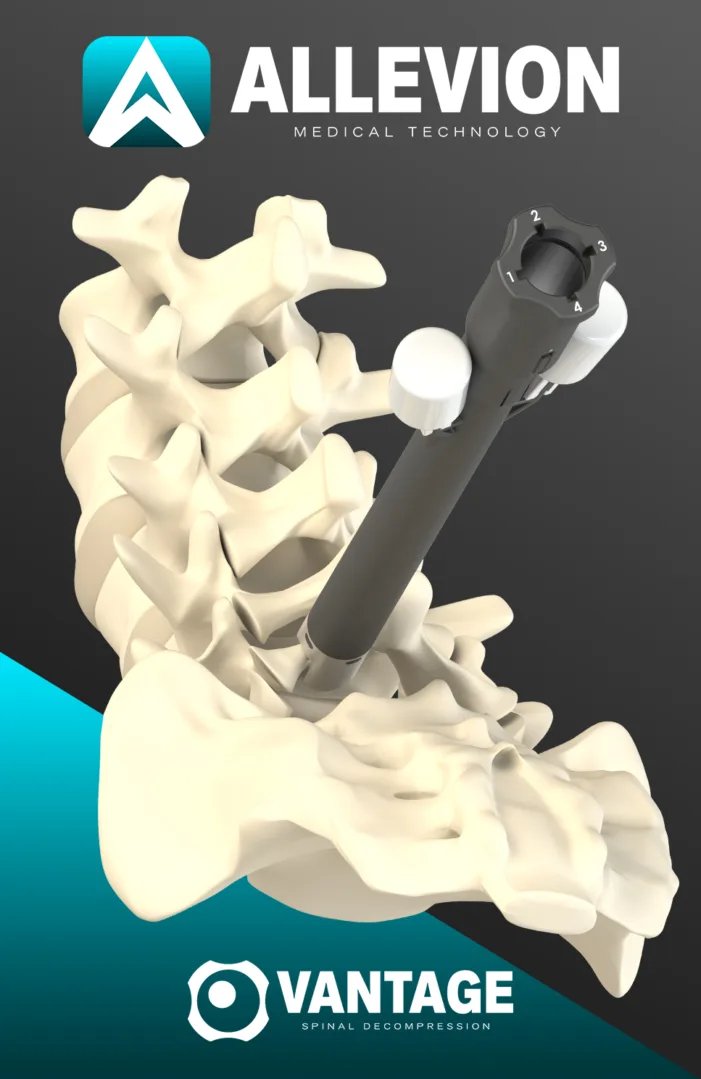
Allevion Medical’s Vantage won FDA 510(k) clearance for lumbar decompression. Read what this may change for spinal stenosis workflow, adoption, and risk.