Why Indivior’s Delaware redomiciliation matters more than a corporate address change

Find out how Indivior’s move to the United States reshapes governance, regulation, and investor perception in addiction treatment biotech.

Find out how Indivior’s move to the United States reshapes governance, regulation, and investor perception in addiction treatment biotech.

Discover why Standard Process’ Cultivate symposium aims to close clinical education gaps in whole-food nutrition and reshape healthy aging care for practitioners.

GenSight Biologics S.A.’s GS010/LUMEVOQ rollout in France and Israel has done more than expand access—it may be redefining how gene therapies are delivered in ophthalmology. As the company prepares for bilateral administration under early access programs for Leber Hereditary Optic Neuropathy (LHON), it is stepping into a growing shift across the field: a move away […]
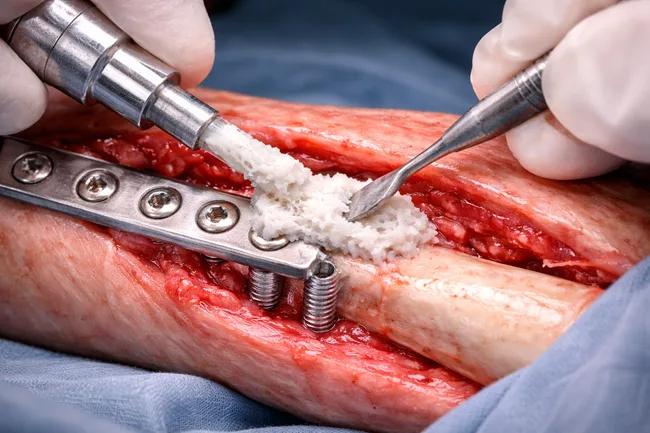
The recent U.S. Food and Drug Administration (FDA) approval for RevBio Inc. to begin a pilot trial of its regenerative bone adhesive, TETRANITE, in dental ridge augmentation has reignited industry attention around a long-elusive biomaterials category: viable, clinically integrated bone adhesives. While dental applications are the entry point, the real strategic question is whether these […]
GenSight Biologics S.A.’s early access rollout of GS010/LUMEVOQ in France and Israel has drawn new attention to how regulators are quietly repurposing compassionate use frameworks into strategic regulatory sandboxes for advanced therapies. As rare disease gene therapies edge closer to commercial viability, a patchwork of early access programs across key jurisdictions is beginning to influence […]
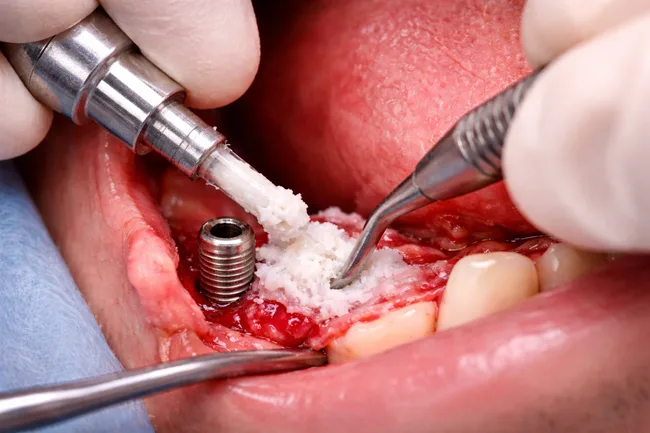
RevBio Inc. has received approval from the United States Food and Drug Administration to initiate a pilot clinical trial evaluating TETRANITE, its regenerative bone adhesive, for dental ridge augmentation procedures. The trial will examine whether the product can eliminate the need for membranes, meshes, tacks, and fixation screws—elements that have traditionally been considered essential for […]

Sanofi’s acquisition of CpG 1018 brings TLR9 adjuvants to the forefront. Explore how this could transform combo vaccines and adult immunization strategies.
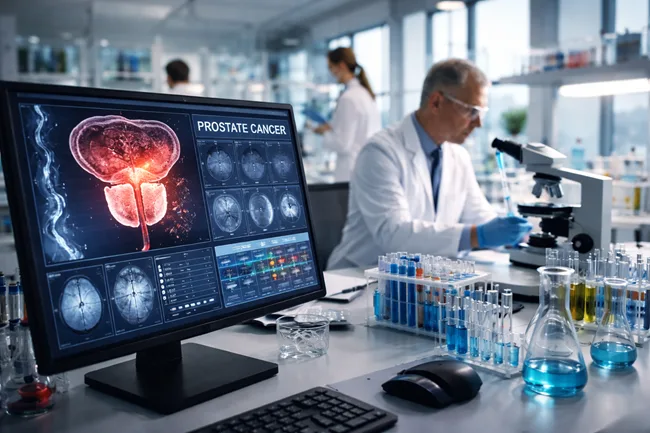
The recent FDA approval of Rubraca (rucaparib) for pre-chemotherapy use in BRCA-mutated metastatic castration-resistant prostate cancer marks a strategic inflection for the field. This expanded indication, driven by head-to-head superiority over docetaxel in the TRITON3 Phase 3 trial, positions BRCA mutations as the first molecular biomarker in prostate cancer to carry actionable, label-defining weight in […]

Discover how biotech startups are targeting the regulome—the cell’s regulatory code—to unlock new drug targets beyond the genome and proteome.

For much of the past two decades, monoclonal antibodies have dominated modern drug development. Their ability to bind with high specificity, block protein function, and engage immune mechanisms has transformed the treatment of cancer, autoimmune disorders, and infectious disease. But despite their blockbuster status, biologics have a blind spot. They are largely excluded from the […]