Faster Candida auris identification? Thermo Fisher Scientific unveils new agar

Thermo Fisher Scientific launches chromogenic media to detect Candida auris faster. Explore what this means for labs and AMR strategy.

Thermo Fisher Scientific launches chromogenic media to detect Candida auris faster. Explore what this means for labs and AMR strategy.

Life Recovery Systems (LRS), a medical device developer headquartered in Alexandria, Louisiana, has received an Investigational Device Exemption (IDE) from the U.S. Food and Drug Administration to advance its ThermoSuit System into a pivotal clinical trial enrolling ischemic stroke patients. The study, which will enroll up to 160 patients across as many as eight hospitals, […]
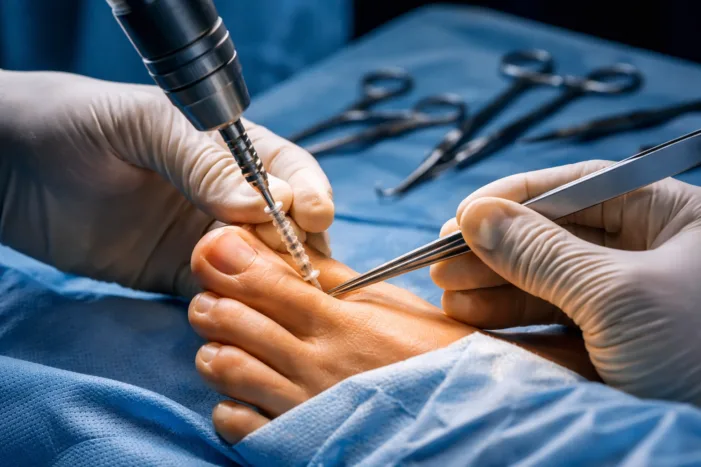
Ossio Inc. launches OSSIOfiber MIA bunion system. Explore what this means for metal free fixation and orthopedic surgery adoption trends.
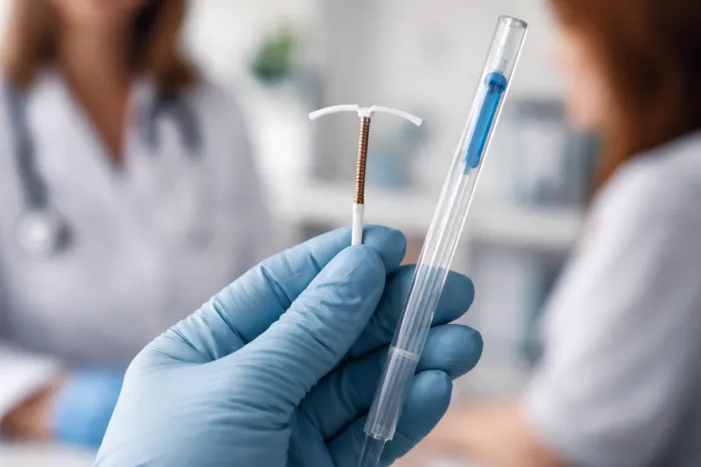
Organon & Co. licenses MIUDELLA copper IUD. Explore what this means for hormone-free contraception and LARC competition. Read the analysis.

Biomerica reports real-world IBS responder data and Medicare coverage. Explore what this means for adoption, reimbursement, and competition.

ClearNote Health’s updated Avantect test improves pancreatic cancer detection in high-risk patients. Find out what this changes for early screening strategies.

Bristol Myers Squibb reports positive luspatercept data in alpha thalassemia. Find out what this could change for treatment and regulation.

20/20 BioLabs launches AI-powered OneTest for longevity risk modeling. Discover what this means for preventive diagnostics and regulation.
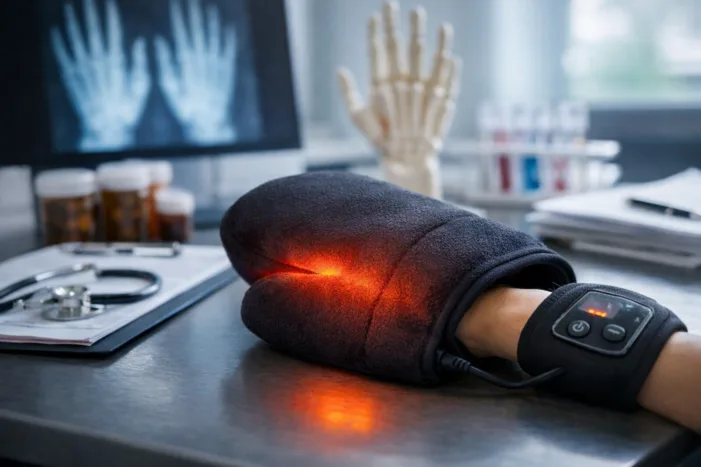
InventHelp’s heat therapy mitten targets hand pain, but what does it really change? Explore the evidence, regulatory gaps, and limits of consumer pain devices.
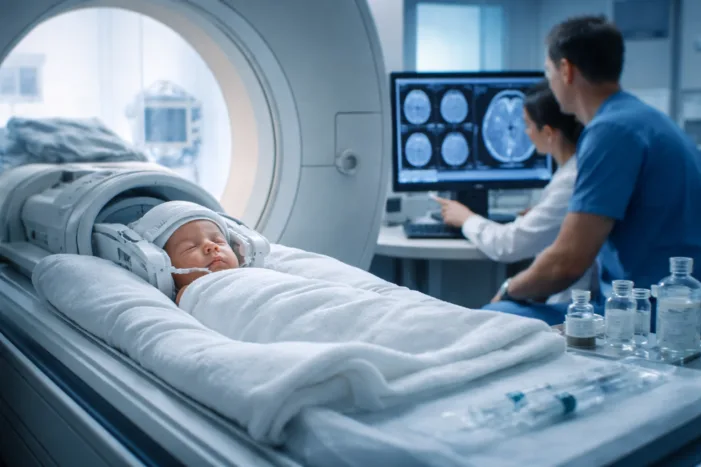
FDA approval expands Guerbet’s gadopiclenol into neonates, reinforcing half-dose MRI contrast strategies. Find out what this changes for pediatric imaging.