Could five-year survival data position KIMMTRAK as a durable standard in uveal melanoma care

Discover how Immunocore’s five-year KIMMTRAK data could reshape survival expectations in metastatic uveal melanoma. Read the full analysis.

Discover how Immunocore’s five-year KIMMTRAK data could reshape survival expectations in metastatic uveal melanoma. Read the full analysis.
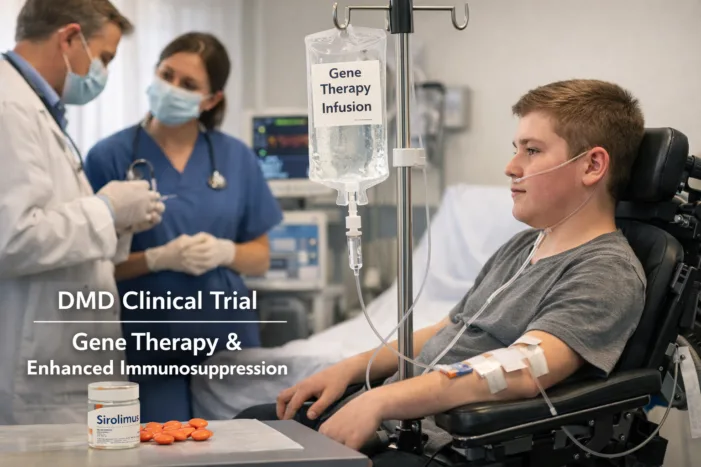
Sarepta’s ELEVIDYS Cohort 8 targets non-ambulatory Duchenne patients with a sirolimus regimen. What the trial design reveals about the path to label restoration.
Aptar’s nasal drug delivery system enters Phase II trial for LTR Pharma’s SPONTAN erectile dysfunction therapy. Discover what this means for intranasal medicines.

Argo Biopharmaceutical Co., Ltd. (Argo Biopharma), a Shanghai-based clinical-stage RNA interference therapeutics company, has received Fast Track Designation from the U.S. Food and Drug Administration for BW-20805, its investigational small interfering RNA therapy targeting prekallikrein for the prevention of hereditary angioedema attacks. The designation comes as the company runs a global Phase II open-label study […]

Alzamend Neuro, the Nasdaq-listed clinical-stage biopharmaceutical company, has initiated a Phase II clinical trial of AL001, its ionic cocrystal lithium formulation, in patients diagnosed with bipolar disorder type 1, conducting the study at Massachusetts General Hospital in collaboration with Harvard Medical School investigators. The trial employs a crossover design in which participants receive both AL001 […]

Sentynl Therapeutics Inc., the United States-based rare disease subsidiary of Zydus Lifesciences Limited, has entered into a licensing agreement with South Korean biotechnology company PRG S&T to develop the investigational molecule Progerinin (SLC-D011) for Hutchinson-Gilford Progeria Syndrome, an ultra-rare genetic disorder that causes accelerated aging in children. The therapy has received orphan drug designation from […]

Meiji Seika Pharma initiates Phase I of ME3241, a PD-1 agonist antibody for autoimmune disease in Australia. Read the clinical analysis.

FDA approves Novartis Cosentyx for adolescent hidradenitis suppurativa. Discover what this decision means for biologic treatment strategies.

Immutep Limited halts its TACTI-004 lung cancer trial after a futility analysis. Discover what the setback means for eftilagimod alfa and LAG-3 immunotherapy.
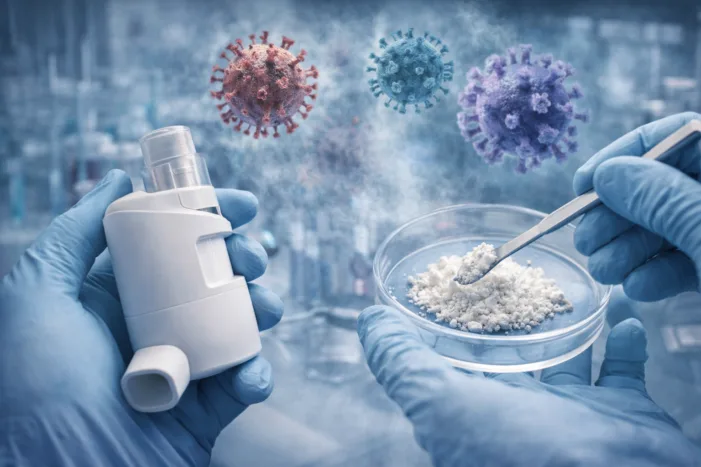
Aptar Pharma and COVIRIX Medical announce a DPI feasibility collaboration for inhaled broad-spectrum antivirals. Read the analysis.