Becton, Dickinson and Company has received CE Mark approval for the Revello vascular covered stent, an endovascular device designed to treat atherosclerotic lesions in the common and external iliac arteries in patients with peripheral artery disease. The approval allows commercialization of the device across CE-mark-accepting European countries, while the technology remains investigational in the United States pending further regulatory evaluation.
The regulatory milestone positions the U.S.-based medical technology manufacturer to enter a competitive but evolving segment of peripheral vascular intervention, where clinicians continue to seek durable solutions for iliac artery disease. While iliac interventions are among the more technically successful procedures in peripheral artery disease treatment, restenosis risk, device durability, and procedural complexity remain persistent challenges that device developers continue to address.
Why BD’s Revello stent approval highlights growing innovation pressure in peripheral artery disease treatment
Peripheral artery disease remains a major global vascular condition affecting hundreds of millions of patients, with prevalence rising sharply in aging populations. Iliac artery disease represents a key subset of these cases, often presenting with symptoms such as claudication and reduced blood flow to the lower extremities. Because the iliac arteries are relatively large vessels compared with femoropopliteal segments, endovascular therapy is often preferred over open surgery.
Industry observers note that the iliac artery segment has historically been one of the more reliable territories for stenting, with high procedural success rates and durable outcomes. However, clinicians still face challenges when lesions are calcified, long, or involve vessel bifurcations. Covered stent technology has therefore gained traction as a potential strategy to improve long-term patency and reduce complications such as plaque prolapse.
The CE Mark approval of the Revello vascular covered stent therefore reflects more than a routine product launch. It underscores continued innovation in device engineering aimed at addressing the anatomical and mechanical stresses unique to iliac arteries, which must accommodate both high radial force requirements and vessel flexibility during patient movement.
How design choices in the Revello device reflect evolving priorities in endovascular engineering
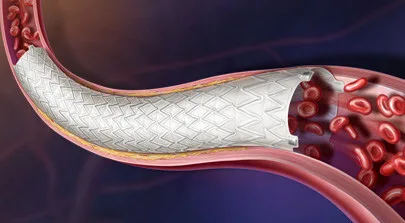
The Revello vascular covered stent combines a nitinol self-expanding scaffold with an ultrathin expanded polytetrafluoroethylene covering, a configuration designed to maintain vessel patency while conforming to anatomical variation. The device also incorporates compliant stent ends intended to reduce trauma to healthy vessel segments and tantalum markers to improve fluoroscopic visibility during deployment.
These engineering features reflect broader trends in endovascular device design. Nitinol remains a widely used material for vascular stents because of its shape memory and superelastic properties, which help maintain vessel scaffolding even under repeated mechanical stress. However, nitinol alone does not address plaque embolization risks or vessel wall coverage, which is where covered stent designs enter the picture.
The use of expanded polytetrafluoroethylene coverings attempts to combine the benefits of a stent with the characteristics of a vascular graft. In theory, this can help seal plaque or thrombus against the vessel wall, potentially reducing restenosis risk in complex lesions.
However, covered stents can also introduce trade-offs. Their presence may obstruct side branches or complicate future interventions. For this reason, clinicians typically select them for specific lesion types rather than as universal replacements for bare metal stents.
What the AGILITY clinical trial may reveal about the device’s long-term clinical relevance
A critical unanswered question surrounding the Revello vascular covered stent involves the clinical data that will ultimately support its long-term use. The device is currently being evaluated in the AGILITY clinical trial, a prospective multicenter study designed to assess performance in patients with peripheral artery disease.
Clinical trial evidence will play an essential role in determining how the device is positioned within treatment algorithms. In peripheral vascular intervention, outcomes such as primary patency, target lesion revascularization, and freedom from major adverse events remain the most closely monitored endpoints.
Regulatory watchers note that European CE Mark approvals under the Medical Device Regulation framework often require less extensive clinical data than U.S. Food and Drug Administration approvals. As a result, post-market evidence generation frequently becomes an important component of device validation in Europe.
If the AGILITY trial demonstrates strong patency rates and favorable safety outcomes, the Revello stent could gain broader adoption among vascular specialists treating iliac artery disease. Conversely, if clinical benefits appear incremental compared with existing devices, adoption could remain limited to specific anatomical scenarios.
How the Revello stent fits within BD’s broader peripheral vascular strategy
For Becton, Dickinson and Company, the Revello stent launch represents a strategic extension of its peripheral vascular portfolio. The Franklin Lakes, New Jersey-based company has increasingly focused on interventional technologies that support minimally invasive procedures, a segment that continues to grow as healthcare systems prioritize shorter hospital stays and reduced procedural complications.
Peripheral vascular devices represent a particularly competitive category within the medical device industry. Major players including Boston Scientific Corporation, Abbott Laboratories, and Medtronic plc maintain strong presences in the endovascular intervention market.
In this environment, differentiation often hinges on incremental engineering improvements, delivery system refinements, and physician familiarity with specific platforms. The Revello stent’s tri-axial delivery system, which is designed to allow controlled deployment and accurate placement, appears intended to address procedural control during complex iliac interventions.
Clinicians tracking device innovation frequently emphasize that ease of deployment can influence adoption as strongly as long-term clinical outcomes. Devices that allow precise placement with fewer procedural steps may gain traction in busy catheterization laboratories where procedural efficiency remains a priority.
Why iliac artery interventions remain an important segment within peripheral vascular care
Although much attention in peripheral artery disease research focuses on femoropopliteal lesions and below-the-knee interventions, iliac artery disease remains a clinically significant condition.
The iliac arteries serve as the primary conduits delivering blood to the lower limbs. When atherosclerotic plaque narrows these vessels, patients may experience severe limitations in walking capacity and reduced quality of life. In advanced cases, compromised blood flow can contribute to limb-threatening ischemia.
Endovascular therapy has become the dominant treatment approach for most iliac artery lesions. Compared with open surgical bypass, minimally invasive interventions typically offer faster recovery and lower complication rates. As a result, device innovation in this segment continues to attract attention from both clinicians and manufacturers.
The Revello stent’s CE Mark approval arrives at a time when European healthcare systems are facing increasing demand for peripheral vascular procedures. Aging populations and rising prevalence of diabetes and cardiovascular disease are expected to expand the patient pool requiring intervention.
What adoption challenges could still limit widespread uptake of the Revello stent
Despite the regulatory milestone, several factors will determine whether the Revello stent achieves meaningful clinical adoption.
First, physicians often rely heavily on devices with long track records. Many iliac stents already have substantial real-world evidence supporting their durability, making clinicians cautious about switching platforms without compelling data.
Second, reimbursement dynamics across European healthcare systems can influence device selection. Hospitals frequently weigh cost considerations alongside clinical performance, particularly in publicly funded systems where budget constraints shape procurement decisions.
Third, procedural familiarity plays a significant role. Interventional specialists often develop strong preferences for specific device platforms based on years of experience. Introducing a new system therefore requires training, physician education, and clear clinical advantages.
Industry observers suggest that early clinical outcomes and physician feedback following the European launch will be closely monitored to assess whether the Revello stent can secure a meaningful foothold in the iliac intervention market.
What regulators, clinicians, and device developers will watch next in the iliac stent landscape
Looking ahead, the success of the Revello vascular covered stent may ultimately depend on how effectively it addresses unresolved challenges in peripheral artery disease treatment.
Clinicians will watch for evidence demonstrating durable patency rates and low complication rates across diverse patient populations. Regulators will evaluate whether ongoing clinical trials support broader approvals, including potential future regulatory submissions in the United States.
Device developers, meanwhile, will continue exploring new engineering strategies aimed at improving vascular interventions. These include thinner stent struts, drug-eluting coatings, and advanced delivery systems designed to enhance precision and minimize vessel trauma.
Within this broader innovation landscape, the Revello stent represents another step in the gradual evolution of endovascular therapy. Whether it becomes a widely adopted standard or remains a niche option will depend on the clinical data emerging in the years ahead.