Artivion’s Endospan acquisition puts NEXUS at the centre of aortic arch device competition

Artivion now owns NEXUS. The bigger test is whether FDA approval can become a scalable U.S. aortic arch business.

Artivion now owns NEXUS. The bigger test is whether FDA approval can become a scalable U.S. aortic arch business.
Artivion’s aortic portfolio is getting stronger, but its 2026 outlook is getting weaker. Can Endospan turn strategy into growth?
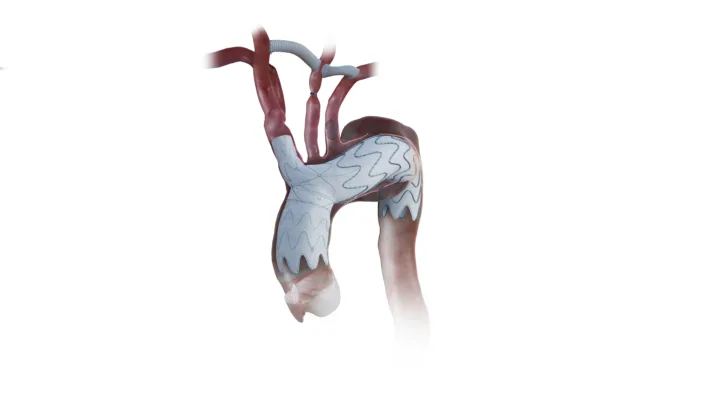
Endospan’s FDA-approved NEXUS system could reshape high-risk aortic arch repair. Read what the approval changes and what clinicians will watch next.
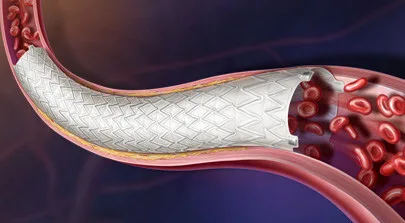
BD secures EU CE Mark for the Revello vascular covered stent for iliac artery disease. Explore what this device could change for PAD treatment in Europe.

Profusa secures Lumee Oxygen orders ahead of EU MDR clearance. Discover what this means for CLTI monitoring and 2026 commercialization.
Philips expands LumiGuide AI 3D light-based navigation across US and Europe. Find out how it could reshape image-guided therapy without radiation.