Can IBI3002 help Innovent Biologics challenge the next wave of type 2 inflammation therapies?

Bispecific antibodies are moving beyond oncology. Innovent’s autoimmune pipeline now faces the harder test of clinical durability.

Bispecific antibodies are moving beyond oncology. Innovent’s autoimmune pipeline now faces the harder test of clinical durability.
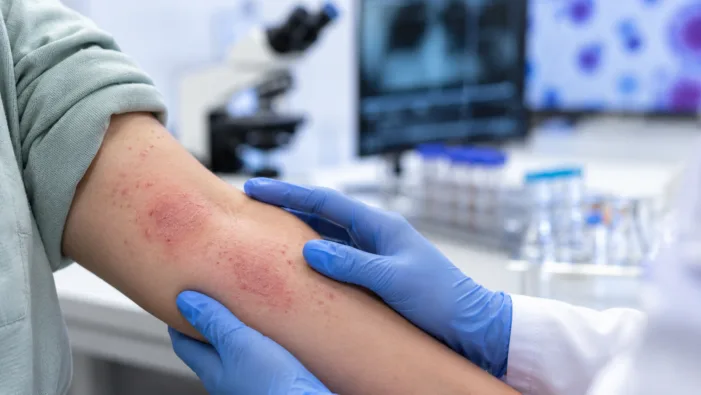
Atopic dermatitis has more drugs, but not enough durable control. TRexBio’s TRB-061 tests whether tissue Tregs can change that.

Can AlloNK make autoimmune cell therapy scalable? Explore why Artiva Biotherapeutics, Inc.’s rheumatoid arthritis strategy matters.

Explore how CPTx’s targeted lipid nanoparticle platform could reshape CAR T therapy scalability and access. Read the full analysis now.

Can CTA313 redefine lupus treatment with CAR-T? Explore what Imviva Biotech’s early data means for autoimmune therapy innovation.

Biomolecular Holdings LLC secures platform patents for tetrahedral antibodies. Discover what this means for the future of antibody engineering.

FDA expands Tzield use to young children with type 1 diabetes. Discover what this means for early intervention and long-term outcomes.

Cue Biopharma, Inc. unveils new CUE-401 data. Find out what this means for autoimmune therapy development and the regulatory path ahead.

Hinge Bio, Inc. doses first subjects in HB2198 Phase 1 trial. Discover what this could mean for lupus treatment and next-generation immune reset therapies.

Innovent Biologics unveiled preclinical IBI3055 data in autoimmune disease. Read what this could change for immune-reset therapies.