Why Cornerstone Robotics’ Sentire CE Mark could shake up Europe’s surgical robotics market
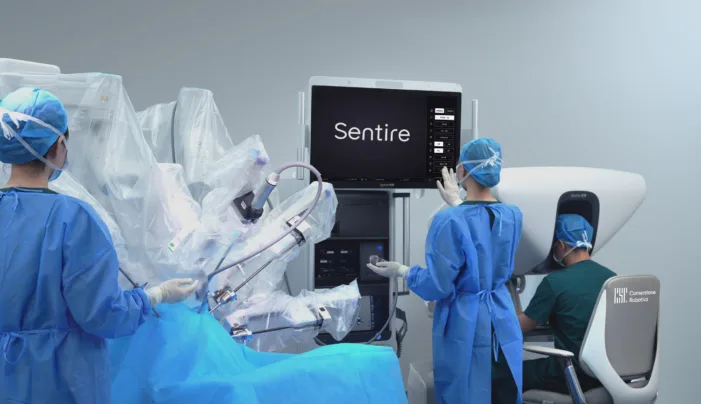
Cornerstone Robotics has cleared Europe’s MDR bar for Sentire. The real test is whether surgical robotics can scale beyond elite hospitals.

Cornerstone Robotics has cleared Europe’s MDR bar for Sentire. The real test is whether surgical robotics can scale beyond elite hospitals.
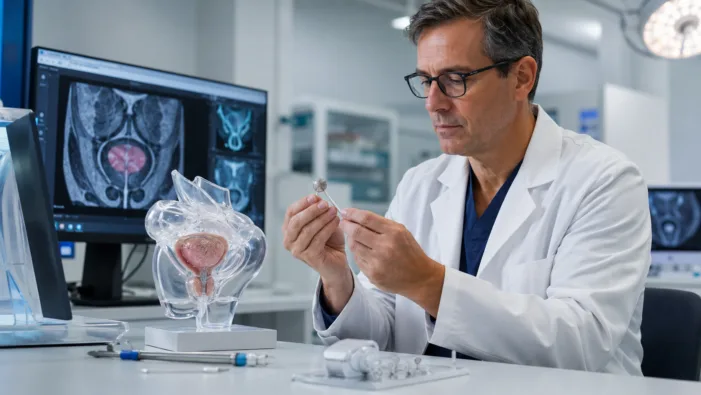
BPH care needs options beyond pills and surgery. Butterfly Medical’s $21m raise puts its reversible prostate implant in the spotlight.

ECMO transport is a clinical weak point. Medtronic’s VitalFlow CE mark tests whether device mobility can strengthen critical care networks.
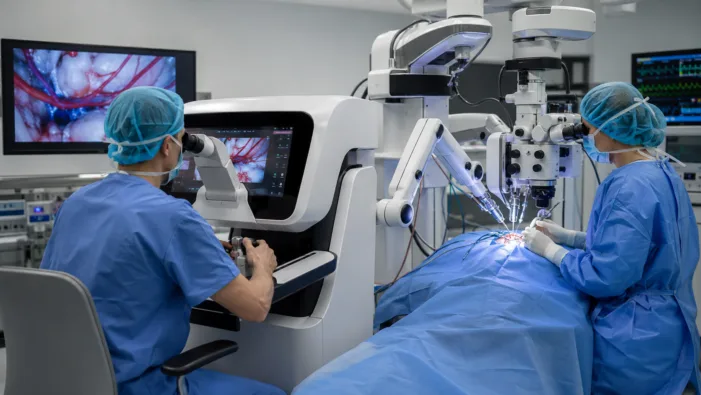
Microsure has European clearance for MUSA-3. Now hospitals must decide whether robotic microsurgery can justify adoption.
Read how Johnson & Johnson’s ETHICON 4000 CE Mark approval could reshape surgical stapling, hospital economics, and future robotic integration.

Profusa secures Lumee Oxygen orders ahead of EU MDR clearance. Discover what this means for CLTI monitoring and 2026 commercialization.
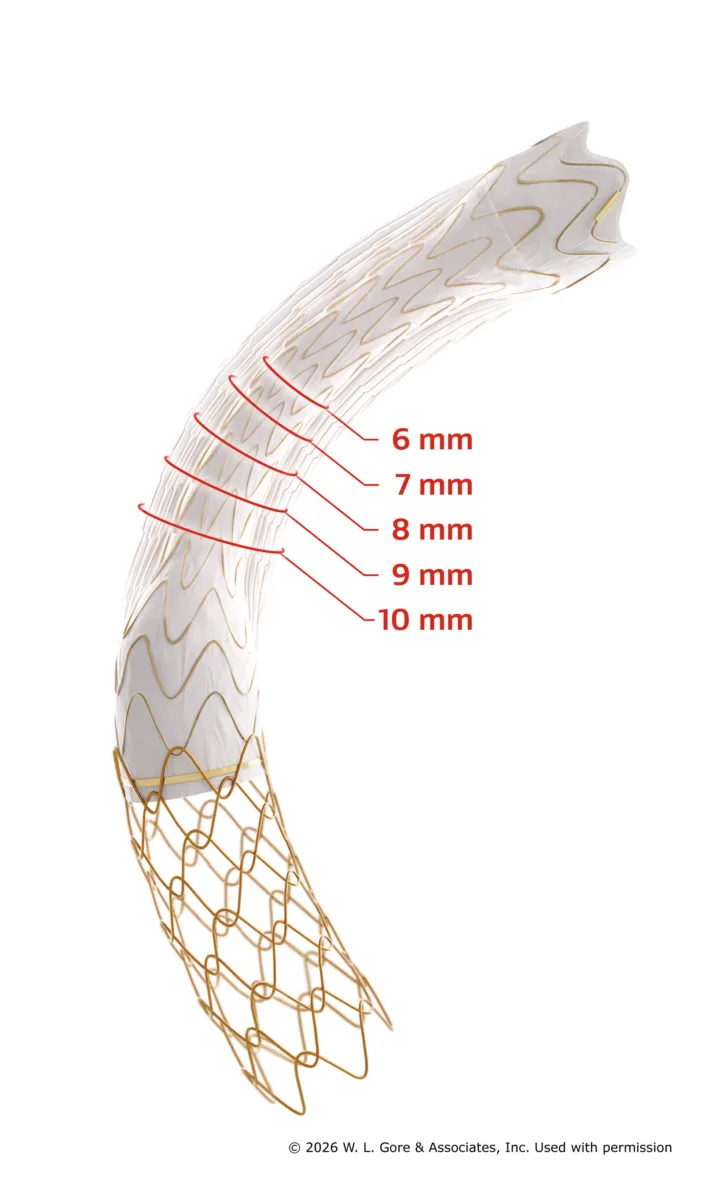
Gore’s VIATORR device earns CE mark for 6–10 mm range, enabling tailored TIPS procedures. Find out how this could reshape portal hypertension care.

Medtronic’s Sphere-360 catheter earns CE Mark and begins U.S. IDE trial. Find out what this means for the future of pulsed field ablation.

May Health’s Anavi System wins CE Mark. Find out what this means for PCOS infertility care, clinical adoption, and regulatory pathways in Europe.

Nitinotes Ltd. has begun patient enrollment in its pivotal U.S. Investigational Device Exemption study evaluating the EndoZip Automated Suturing System for endoscopic sleeve gastroplasty (ESG). The first procedure in the EASE Clinical Trial was completed at Lenox Hill Hospital | Northwell Health in New York, marking the U.S. debut of the device. This study pits […]