Why Harmony Biosciences’ pitolisant approval changes the risk calculus in pediatric narcolepsy

FDA approval expands pitolisant into pediatric cataplexy. Explore what this changes for narcolepsy care, safety, and long-term strategy.

FDA approval expands pitolisant into pediatric cataplexy. Explore what this changes for narcolepsy care, safety, and long-term strategy.
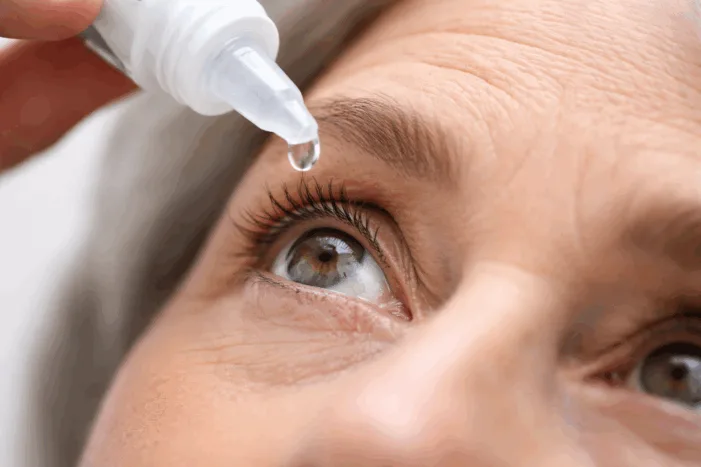
YUVEZZI becomes the first FDA-approved dual-agent eye drop for presbyopia. Find out what this means for Tenpoint Therapeutics and the future of vision care.

Find out how DARZALEX FASPRO’s FDA approval for transplant-ineligible myeloma patients could shift frontline treatment standards.

Sarepta’s gene therapy ELEVIDYS shows 3-year durability in Duchenne muscular dystrophy. Find out what it means for patients, payers, and global rollout.
Organon’s etonogestrel implant NEXPLANON has received FDA approval for extended use up to five years, marking a significant regulatory win for the women’s health–focused pharmaceutical company. The decision, which comes via a supplemental new drug application (sNDA), builds on existing data supporting the implant’s high efficacy and safety profile and broadens its potential utility among […]

FDA approves NEXPLANON for five years of use with REMS program. Find out how this reshapes contraceptive access, safety, and policy impact.
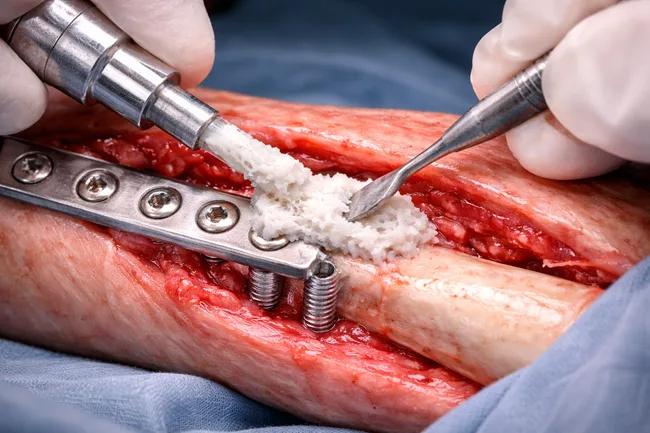
The recent U.S. Food and Drug Administration (FDA) approval for RevBio Inc. to begin a pilot trial of its regenerative bone adhesive, TETRANITE, in dental ridge augmentation has reignited industry attention around a long-elusive biomaterials category: viable, clinically integrated bone adhesives. While dental applications are the entry point, the real strategic question is whether these […]
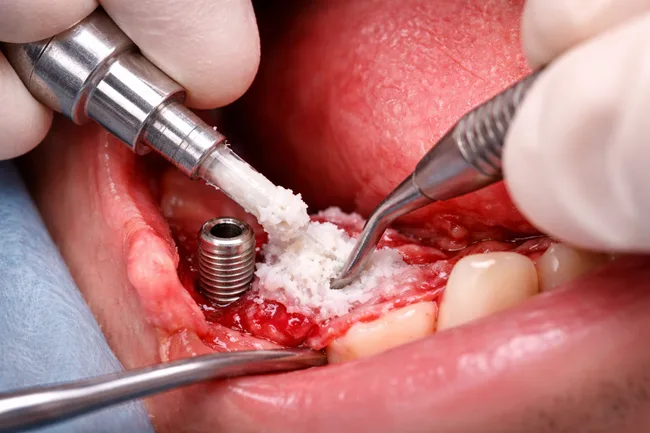
RevBio Inc. has received clearance from the United States Food and Drug Administration to begin a pilot clinical trial of TETRANITE, its regenerative bone adhesive, for dental ridge augmentation. The study will assess the product’s ability to support bone grafting procedures without the use of membranes, meshes, tacks, or screws—offering a potential simplification to a […]

Edwards Lifesciences’ SAPIEN M3 becomes the first FDA-approved transseptal mitral valve replacement system. Explore how it reshapes TMVR in 2026.
FDA approves YARTEMLEA for TA-TMA. Find out how Omeros’ new drug could change transplant care for children and adults alike.