Can Samsung Bioepis reshape Europe’s retinal disease biosimilar market with OPUVIZ?

Find out how Samsung Bioepis’ OPUVIZ launch could reshape Europe’s aflibercept biosimilar market and ophthalmology access.

Find out how Samsung Bioepis’ OPUVIZ launch could reshape Europe’s aflibercept biosimilar market and ophthalmology access.

Bispecific antibodies are moving beyond oncology. Innovent’s autoimmune pipeline now faces the harder test of clinical durability.
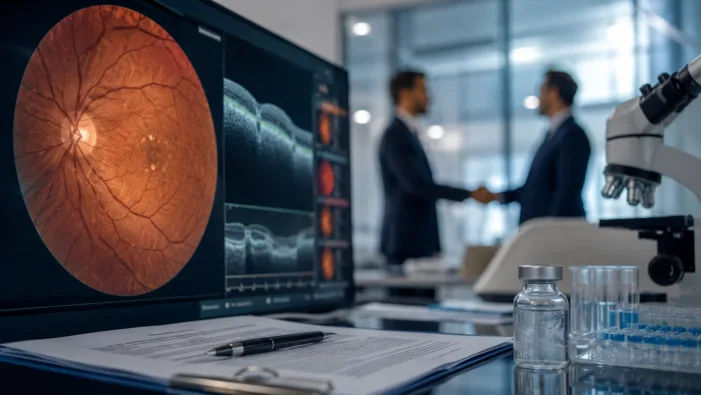
Eylea is under pressure. Bayer’s $2.45bn Perfuse bet tests whether PER-001 can carry its next ophthalmology growth story.

Vision testing is getting faster. PerZeption and Alcon now face the harder test: proving AIM+ CSF can reshape research-grade evidence.
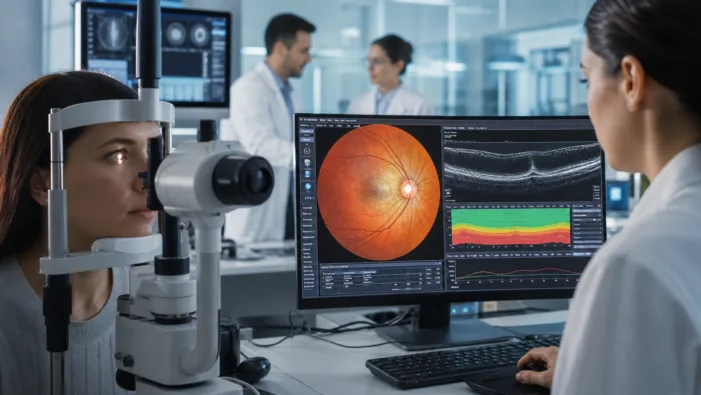
Retinal disease care still targets drivers. Spinogenix is testing whether synaptic regeneration can protect vision when damage continues.
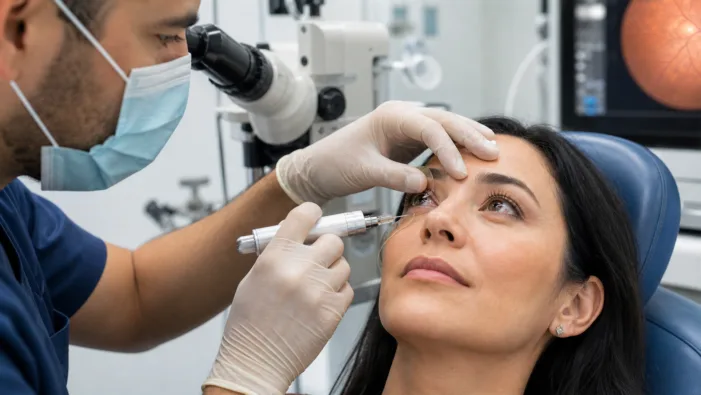
Retina drugs are improving, but delivery remains the bottleneck. Everads Therapy is testing whether suprachoroidal access can change that.
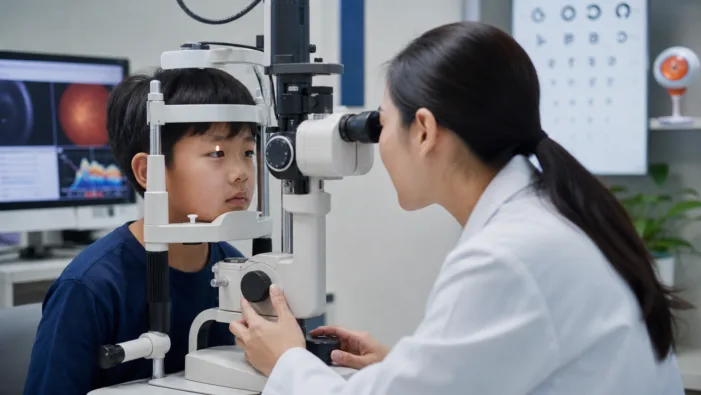
Pediatric myopia is rising fast, but U.S. drug options remain thin. Sydnexis’ SYD-101 data may sharpen the regulatory race.

Glaukos secures a permanent J-code for Epioxa. Read why this reimbursement milestone could shape keratoconus treatment adoption and payer access.
Johnson & Johnson’s TECNIS PureSee data could reshape premium cataract surgery. Read what the ASCRS 2026 findings may change next.

Glaukos heads to ASCRS 2026 with Epioxa and glaucoma data. Read what this could change for keratoconus, sustained therapy, and adoption.