Trisol Medical, a clinical-stage device firm based in Israel, has released positive results from its U.S. Early Feasibility Study evaluating its Transcatheter Tricuspid Valve Replacement (TTVR) system in patients with severe to torrential tricuspid regurgitation (TR). The system, tested via trans-jugular access in 22 high-risk patients, demonstrated safety, functional improvement, and successful deployment, including in those with impaired right ventricular function—a subgroup often excluded from early-stage structural heart trials.
The announcement marks a critical step forward for the emerging TTVR category, where procedural complexity, anatomical variability, and high comorbidity burdens have historically slowed clinical and commercial progress.
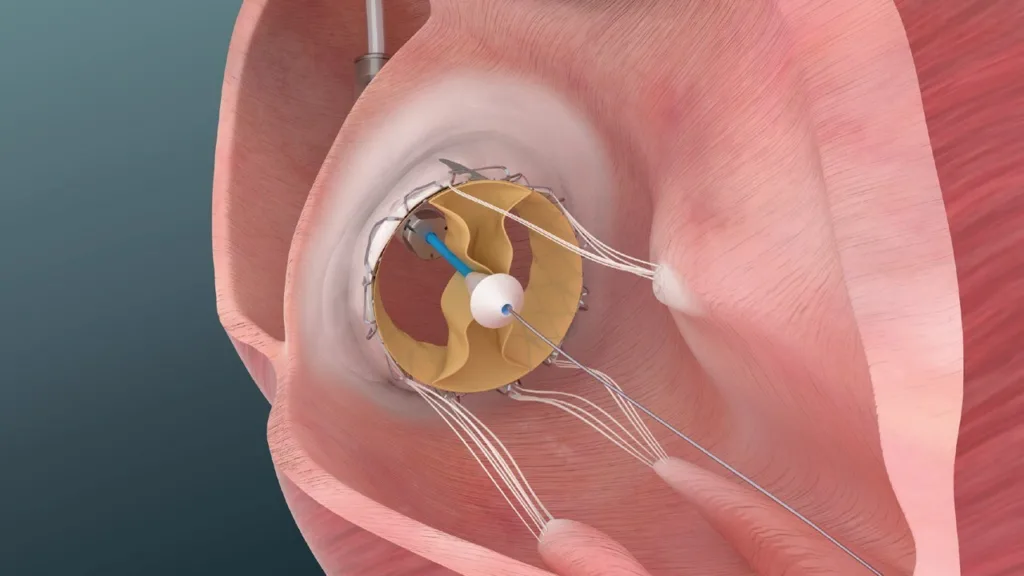
Why Trisol’s data may reframe expectations in the underdeveloped tricuspid replacement market
The most notable aspect of Trisol Medical’s feasibility results is not simply that the device worked in a difficult population, but that the early safety and functional outcomes challenge existing assumptions about right ventricular impairment as an exclusionary factor. Historically, reduced right ventricular function has been a confounding variable in both trial design and real-world applicability for transcatheter tricuspid repair or replacement. While competing systems—particularly those pursuing edge-to-edge repair—have often struggled to demonstrate efficacy in this group, Trisol’s device showed quality-of-life improvement and cardiac output gains at 30-day and 12-month follow-ups.
This strengthens the argument that tricuspid regurgitation should be addressed more aggressively even in advanced patients rather than being considered terminal or too risky for intervention. The use of Kansas City Cardiomyopathy Questionnaire (KCCQ), NYHA class improvement, six-minute walk distance, and hemodynamic parameters adds multi-dimensional clinical relevance to the data, though long-term durability and broader applicability remain to be demonstrated in pivotal trials.
What makes the Trisol valve structurally and procedurally distinct from other TTVR contenders
Technologically, the Trisol system appears to bring several differentiated features to a category still in flux. While much attention in structural heart innovation has focused on aortic and mitral transcatheter platforms, the tricuspid valve remains structurally complex and challenging to treat due to its size, lack of calcification for anchoring, and variability in anatomy. Trisol’s valve design, featuring recapturable anchors and a broad size range, appears engineered with these specific anatomical challenges in mind.
Perhaps more importantly, early data shows a permanent pacemaker implantation rate of less than 5% at 30 days, a figure that compares favorably with rates observed in transcatheter mitral and tricuspid repair trials. Low pacemaker dependency has increasingly become a key commercial and procedural threshold for interventional cardiology adoption, particularly in a frail elderly cohort. A successful shift to transfemoral access could further align Trisol with workflow preferences across top centers globally.
Industry observers note that the ability to deploy the device without migration, combined with a stepwise pivot from trans-jugular to transfemoral delivery, could strengthen the platform’s profile as a flexible option for varying operator preferences and patient anatomies. However, independent validation of these outcomes in larger cohorts remains necessary before drawing broader procedural conclusions.
Regulatory clarity remains limited, but Trisol’s pathway mirrors that of mitral innovations
From a regulatory standpoint, Trisol Medical’s system remains in early feasibility stages in the United States, a process that allows for protocol adaptation and enrollment modifications but offers limited predictive power regarding full market approval. The U.S. Food and Drug Administration (FDA) has in recent years shown flexibility in advancing novel heart valve technologies through its Breakthrough Devices Program, but final pathway acceleration often depends on consistency across centers, durability endpoints, and comparability to either surgical benchmarks or medical therapy in randomized arms.
The broader transcatheter tricuspid landscape includes multiple systems in various stages of clinical development, including orthotopic replacement devices, heterotopic caval implants, and edge-to-edge repair platforms. However, no TTVR system has yet secured full FDA approval. Industry watchers suggest that as a second-wave TTVR system, Trisol may benefit from regulatory learnings derived from mitral and aortic pathways but will still need to build a differentiated risk-benefit narrative given the fragility and complexity of the TR population.
Adoption and scalability challenges will depend on operator experience and health economics
Despite encouraging feasibility data, Trisol Medical’s commercial viability will ultimately depend on adoption dynamics and procedural scalability. For one, the learning curve for transcatheter tricuspid valve replacement remains steep. Many high-volume centers are still in the early stages of building experience with tricuspid anatomy and procedural planning. Devices requiring significant imaging support or anatomical customization may face adoption bottlenecks outside of elite structural heart programs.
Moreover, while the initial study was conducted in high-risk surgical patients, reimbursement frameworks will require evidence of superiority or at least non-inferiority against conservative medical management in broader risk categories. Payers are expected to scrutinize the cost-benefit ratio of TTVR devices in elderly, co-morbid patients with limited life expectancy or frail baseline status. Longitudinal data on rehospitalization rates, device durability, and heart failure progression will be key to making the reimbursement case.
Clinicians tracking the field believe that successful expansion to a transfemoral delivery route could meaningfully reduce procedural complexity, improve recovery timelines, and align Trisol with prevailing reimbursement and care delivery models in both the United States and Europe.
What clinicians, trialists, and industry stakeholders are watching as pivotal trials approach
The transition from early feasibility to pivotal trial design represents a major inflection point for Trisol Medical. Stakeholders will be closely monitoring how the company defines its pivotal endpoints—particularly whether it pursues a single-arm safety and performance study or attempts a randomized control trial comparing to standard-of-care medical management. The inclusion of patients with poor right ventricular function will also be a critical test of the device’s real-world impact.
Additionally, emerging comparative data across next-generation TTVR systems will heighten scrutiny on technical success, paravalvular leak rates, and functional gains. While Trisol’s recapturable anchor and broad sizing strategy are attractive, durability and valve thrombosis data over 1–2 years will be crucial to making a case for structural superiority.
Industry observers note that patient segmentation strategies may also evolve. There is growing interest in combining transcatheter tricuspid devices with other therapies such as left ventricular assist devices, electrophysiology interventions, or combined mitral-tricuspid repair in patients with biventricular dysfunction. Whether Trisol can integrate into this broader procedural ecosystem will depend on modularity, valve-in-valve options, and cross-specialty comfort levels.