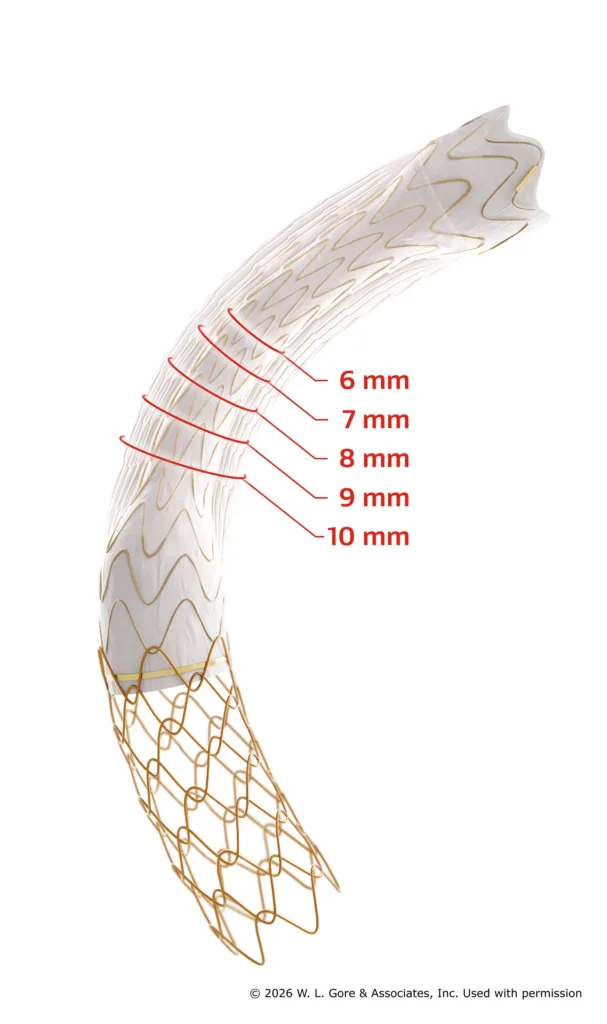
W. L. Gore & Associates Medical Products has secured CE mark approval for a new 6–10 mm diameter configuration of the GORE VIATORR TIPS Endoprosthesis with Controlled Expansion, expanding treatment flexibility for physicians managing portal hypertension. The device is now the first CE-marked transjugular intrahepatic portosystemic shunt (TIPS) prosthesis to offer this full diameter range, advancing Gore’s position in the space of hepatic vascular intervention and reflecting shifting clinical strategies that increasingly favor smaller-caliber, precisely controlled shunt placement.
Why a broader diameter range matters in portal hypertension management strategies
The newly approved range extends the device’s previously available 8–10 mm configuration by introducing a 6 mm starting diameter, addressing a known gap in shunt calibration options. This is not a superficial expansion. Shunt size plays a pivotal role in achieving an optimal portal pressure gradient while minimizing the risk of adverse outcomes such as hepatic encephalopathy, volume overload, and cardiac dysfunction—common complications associated with excessive diversion of portal flow.
Clinicians have long debated the balance between efficacy and safety in TIPS procedures. Smaller-diameter shunts, often associated with fewer systemic effects, have lacked sufficient commercial availability in CE-marked devices with consistent radial control. The controlled expansion capability built into Gore’s device offers a clinical bridge between procedural predictability and patient-specific tailoring. Industry observers believe this configuration shift may allow European physicians to standardize a more conservative shunt strategy without resorting to off-label improvisation or multiple device inventories.
What this signals about shifting clinical attitudes toward low-diameter shunt thresholds
Portal hypertension treatment is increasingly embracing tailored flow modulation over aggressive decompression. This is partly a response to rising concerns around hepatic encephalopathy risk, particularly in cirrhotic patients with borderline neurocognitive profiles. The trend has been supported by multiple studies highlighting that initial 6 mm or 7 mm TIPS placements can reduce symptom burden without triggering excessive cardiac preload.
The endorsement from European regulators adds further weight to this evolving clinical philosophy. While 8–10 mm shunts were historically the norm, gastroenterologists and interventional radiologists tracking outcomes across Europe have indicated growing demand for more granular intraoperative control. By positioning its device as adjustable yet reliable, Gore effectively underlines this evolution.
How controlled expansion tech differentiates Gore from non-modular alternatives
The GORE VIATORR TIPS Endoprosthesis incorporates proprietary expanded polytetrafluoroethylene (ePTFE) graft material, which resists bile and mucin permeation—common causes of stent dysfunction. But the controlled expansion mechanism is the more significant differentiator, particularly when compared with fixed-diameter stents that lack the ability to be fine-tuned during deployment.
This design enables operators to begin with a lower initial diameter and then expand incrementally based on the measured portosystemic gradient. Clinical teams can thus reduce the need for post-deployment ballooning or additional instrumentation, cutting down on procedural variability. Some hospital procurement teams across Germany and Italy have reportedly favored this modularity in device tenders, especially where case-to-case customization is required without incurring extra stock burdens.
Regulatory watchers suggest that this combination of flexibility and long-term patency data could pressure competitors with static-diameter covered stents to reevaluate both product portfolios and trial endpoints.
What this approval changes for inventory management, procedural planning, and reimbursement policies
From a hospital administration standpoint, the expanded diameter range could reduce the number of different SKUs physicians must maintain in catheterization labs. Instead of selecting between multiple fixed-diameter devices, physicians can now use one adjustable product to accommodate variable patient anatomies and pressure targets.
This also aligns well with emerging procurement strategies in European health systems that prioritize cost-efficient, multi-use devices capable of simplifying supply chains. Several reimbursement authorities are in the process of revising device groupings for vascular interventions, and analysts believe modular TIPS devices may qualify for more favorable billing codes tied to clinical adaptability or reduced complication rates.
While U.S. reimbursement remains a distinct regulatory pathway, the CE mark for this device could be interpreted as a proof-of-concept milestone that may bolster future applications for expanded FDA labeling. Whether Gore pursues an updated Premarket Approval (PMA) supplement to reflect the new configuration in the United States remains to be seen, but the European validation will likely serve as key supportive evidence.
Remaining limitations and what clinicians will likely monitor next
Despite its engineering improvements, the device does not eliminate the core challenge of balancing portal decompression with systemic stability. Long-term comparative data for the 6–7 mm range within the VIATORR platform remain limited, particularly across subgroups such as post-transplant patients, alcohol-related cirrhosis, or those with pre-existing cardiac compromise.
In addition, while controlled expansion can mitigate some risks, it places greater responsibility on procedural teams to accurately titrate shunt dimensions in real time. Operator variability and institutional differences in measuring portal pressure gradients could still influence outcomes—an issue flagged in prior multicenter audits of TIPS performance across Europe.
Some interventional specialists also caution that while smaller shunts may lower encephalopathy rates, they may not offer sufficient decompression in cases of refractory variceal bleeding or hepatorenal syndrome, where aggressive gradient reduction is necessary. Tailoring must therefore occur within a broader risk-stratified framework, not as a universal shift toward minimalism.
How this strengthens Gore’s moat in the European vascular intervention market
With this CE mark approval, W. L. Gore & Associates strengthens its leadership in a niche but clinically vital segment of interventional hepatology. The VIATORR platform already holds a dominant position due to its track record of patency, durability, and technical reliability, but the newly approved 6–10 mm variant enhances its adaptability narrative.
For European hospitals consolidating vendors or updating treatment algorithms for portal hypertension complications, the expanded configuration could emerge as a default choice—particularly in centers prioritizing complication reduction and outcome-based care.
Competitor devices without expansion control or flexible sizing will likely struggle to justify comparable value, especially in settings where reimbursement is increasingly tied to procedural precision and complication avoidance. The fact that Gore’s device now accommodates both conservative and standard shunting strategies within a single framework may make it difficult for rivals to counter on flexibility or economic efficiency grounds.
While broader adoption will depend on physician training, procedural standardization, and hospital policy, this CE mark approval marks a pivotal step in the modularization of hepatic vascular therapy.