RevBio Inc. has received approval from the United States Food and Drug Administration to initiate a pilot clinical trial evaluating TETRANITE, its regenerative bone adhesive, for dental ridge augmentation procedures. The trial will examine whether the product can eliminate the need for membranes, meshes, tacks, and fixation screws—elements that have traditionally been considered essential for successful bone grafting prior to dental implant placement. The study represents an inflection point in the evolving category of adhesive-based biomaterials and could redefine the way dental surgeries are performed in high-throughput clinical settings.
Why current ridge augmentation remains hardware-heavy and prone to failure
Conventional ridge augmentation procedures are required in a large subset of dental implant cases where the alveolar ridge has resorbed or collapsed following tooth loss. These procedures typically use particulate bone grafts to rebuild lost bone volume, which then must be protected during healing. This is usually achieved through resorbable membranes or titanium meshes, often anchored in place with tacks or screws to stabilize the graft and exclude soft tissue infiltration.
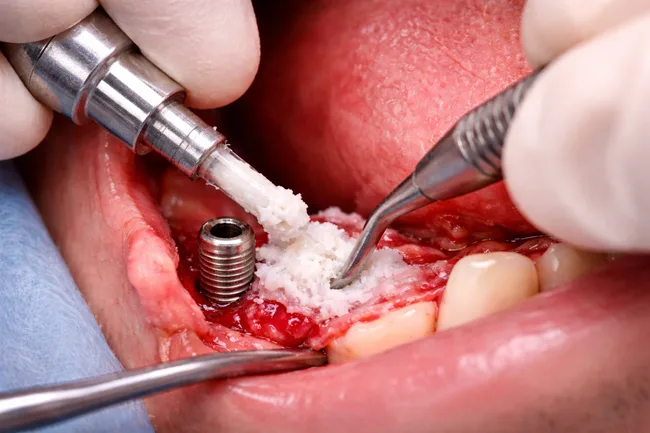
Despite widespread use, these hardware-dependent techniques come with a range of limitations. Resorbable membranes can degrade too quickly or unpredictably, leaving graft material vulnerable to collapse or contamination. Titanium meshes, while more stable, increase the risk of mucosal irritation and frequently require a second procedure for removal. The fixation devices themselves can create complications, from patient discomfort to infection and difficulty in achieving a clean surgical field.
More concerning is the outcome data. Clinical literature and practitioner reports indicate that more than 30 percent of ridge augmentation cases fail to achieve the required bone volume or structural integrity on the first attempt. This often results in additional surgeries, higher treatment costs, extended timelines, and reduced patient satisfaction. The complexity and variability of current methods continue to pose a significant barrier, particularly in general practice or DSO environments where surgical specialization may be limited.
How TETRANITE could collapse three procedural steps into one biomaterial
The central innovation behind TETRANITE lies in its multifunctionality. Developed by RevBio Inc., the adhesive is designed to adhere directly to bony walls, form a temporary scaffold, and resist soft tissue ingrowth. This eliminates the need for separate containment and fixation hardware. The material is bioresorbable and osteoconductive, meaning it gradually integrates with the body while allowing for new bone formation within a clinically relevant timeline.
By consolidating three essential roles—barrier, scaffold, and fixation—into a single biomaterial, TETRANITE presents an opportunity to streamline ridge augmentation into a less invasive, more predictable procedure. The product’s behavior in a wet surgical field, which is notoriously difficult for many biomaterials, is a key differentiator. It cures quickly, maintains its form, and retains adhesive strength even in the presence of saliva or blood.
This could eliminate one of the most complex steps in graft preparation: securing the material. Without the need to shape and affix membranes or mesh using screws or pins, clinicians can reduce chair time, limit variability, and improve procedural efficiency. For patients, this could mean fewer appointments, lower complication risks, and a faster timeline to implant placement.
Why chairside simplification could matter more than material science in the long run
The real disruption TETRANITE may offer lies not just in its clinical performance, but in its workflow potential. High-throughput practices, including multi-site dental service organizations and large implant centers, are increasingly focused on standardizing care to reduce training burdens and enhance profitability. A material that simplifies the surgical protocol, eliminates ancillary tools, and reduces follow-up interventions fits squarely into that operating thesis.
Clinicians and analysts suggest that chairside graft simplification could lead to increased implant conversion rates, especially in patient populations who might otherwise decline treatment due to the added cost, discomfort, or procedural complexity of ridge augmentation. If TETRANITE allows clinicians to graft and stabilize a defect in one step using only a pre-packaged cartridge or molded unit, that could lower the procedural barrier for general dentists and early-career implantologists.
For DSOs, this simplification translates into higher procedural throughput and reduced inventory complexity. Fewer SKU variations, fewer steps in surgical kits, and less dependency on specialist training could all contribute to stronger margins and higher utilization of chair time. Moreover, risk reduction through fewer postoperative complications or graft failures aligns well with the economic incentives of value-based reimbursement models, which are slowly entering the dental space.
How biomimetic bone adhesives compare with PRF, bone putties, and conventional graft materials
TETRANITE does not exist in a vacuum. It enters a market already populated with biologics and engineered materials aimed at improving bone graft outcomes. Platelet-rich fibrin (PRF), for example, is widely used to enhance healing and reduce inflammation, but it lacks the structural rigidity required for ridge augmentation on its own. Bone putties, including those based on calcium phosphate or hydroxyapatite, offer easy handling and moldability but typically do not maintain their volume under compressive forces.
Particulate grafts remain the gold standard but require precise manipulation, containment membranes, and positional stabilization. Without these support elements, particulate material often fails to maintain form, particularly in horizontal ridge defects. This leads to underbuilding and necessitates regrafting in future stages.
TETRANITE, by contrast, is designed to integrate directly with surrounding bone while forming a load-bearing, defect-conforming structure. This positions it closer to a “set-and-heal” solution rather than a “pack-and-protect” one. If the clinical trial validates both osteoconductivity and adhesive resilience, the product could redefine how clinicians select graft materials for smaller, localized augmentation procedures where volume retention and workflow efficiency are more critical than osteoinductive potency.
What RevBio’s trial reveals about broader shifts in surgical biomaterials and regulation
The FDA’s decision to greenlight a first-in-human trial for TETRANITE in dental ridge augmentation is significant for more than just the product’s future. It also reflects a growing regulatory comfort with synthetic, multifunctional biomaterials that challenge conventional product categories. TETRANITE is currently involved in ten different FDA-approved trials spanning neurosurgical, orthopedic, and dental indications. This platform-based approach could ultimately support a De Novo classification for multiple use cases.
The funding support behind the program has also been notable. RevBio Inc. has received $1.8 million in grants from the National Institute of Dental and Cranial Research via the Translational Resource Center, as well as a $2 million Small Business Innovation Research award. These grants reflect not only institutional confidence in the underlying science but also the belief that biomaterials with multifunctional capabilities are critical to advancing regenerative medicine.
As regulators evaluate TETRANITE’s performance, industry observers expect attention will be paid to handling, failure modes, and postoperative follow-up data, not just primary efficacy endpoints. If the material avoids the common pitfalls of graft migration, soft tissue penetration, or premature resorption, it could become one of the first widely adopted synthetic adhesives in dental implantology.
What the clinical community will watch as adhesives move toward mainstream use
From the perspective of clinicians, the upcoming trial will raise several important questions. Chief among them is whether TETRANITE can demonstrate comparable or superior outcomes to traditional ridge augmentation techniques while offering a simplified workflow. Clinicians will also be evaluating the material’s resorption profile, its radiographic visibility during healing, and whether it integrates predictably with subsequent implant placement protocols.
Training and operability will also be key considerations. If the product requires minimal learning curve and fits seamlessly into existing kits or chairside delivery tools, adoption could be swift. However, if the handling properties or application method require retraining or new instrumentation, that could slow uptake in the short term.
From a broader market standpoint, RevBio Inc. is positioning TETRANITE not just as a product, but as a category-opening platform. If it succeeds in the dental space, expansion into craniofacial reconstruction, spinal fusion adjuncts, or trauma repair may follow. But the initial test—whether a glue can reliably substitute for the combined function of bone graft, barrier membrane, and screw fixation—will be closely scrutinized by all stakeholders.
Why procedural convergence may matter more than graft volume in future implant workflows
What RevBio Inc. is ultimately proposing is a rethinking of how clinicians approach hard tissue regeneration. TETRANITE is not trying to outperform every graft material in osteoinduction or remodeling speed. Instead, it is seeking to collapse procedural complexity and improve consistency. If successful, this approach could be more transformative than incremental improvements in any single performance metric.
The move toward adhesive, multifunctional biomaterials fits into a broader trend of surgical convergence—where tools, scaffolds, and barriers are no longer separate but integrated into single-use, targeted delivery formats. Ridge augmentation, long considered a slow and failure-prone step in the implant journey, could become faster, cleaner, and more predictable.
And if that proves true, dental bone glue may not just be a niche innovation. It could be a foundational upgrade in the biomaterial toolkit that shapes the next chapter of graft-free implantology.