Hyundai Bioscience begins U.S. Phase 2 trial of Xafty for COVID, flu, and RSV

Hyundai Bioscience enters U.S. Phase 2 trials for Xafty, a broad-spectrum antiviral targeting flu, RSV, and COVID. Find out what this means for future pandemics.

Hyundai Bioscience enters U.S. Phase 2 trials for Xafty, a broad-spectrum antiviral targeting flu, RSV, and COVID. Find out what this means for future pandemics.

AskBio’s AB-1009 gene therapy for late-onset Pompe secures FDA IND clearance. Find out what this milestone means for AAV innovation and patient outlook.

TheraTears adds eyelid wipes and preservative-free drops for screen fatigue. Find out what this expansion means for OTC dry eye care and consumer trends.

Explore how Biocorneum’s dual-action bruise care system could reshape post-procedure recovery in aesthetic medicine. Read the strategic analysis now.

Find out how AIROS Medical’s five-year warranty on compression devices may shift durability standards and procurement expectations in lymphedema care.

AVEO begins new trial of ficlatuzumab in front-line AML with Beat AML. Find out what this signals for older patients and c-Met inhibitor development.

Lynx Analytics has introduced Lumen, a dedicated agentic artificial intelligence framework designed to support complex decision-making across life sciences workflows. The launch was anchored by a real-world deployment at AstraZeneca, where a custom-built application called IlluminAI is now embedded in the oncology team’s brand planning process across international markets. The framework is positioned as a […]
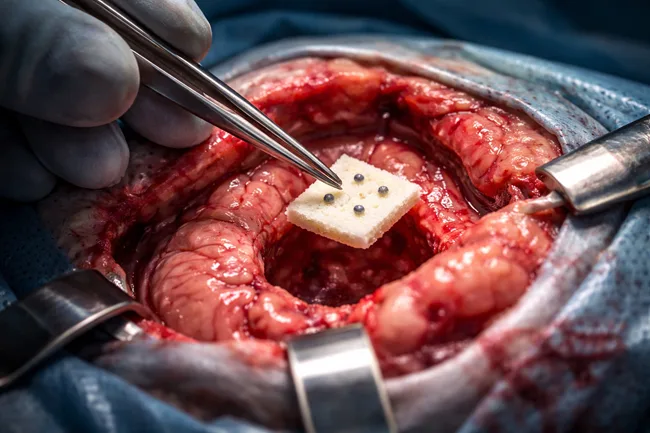
GT Medical Technologies has announced the first patient enrollments in its BRIDGES trial, a randomized clinical study assessing the use of GammaTile therapy in newly diagnosed glioblastoma. The trial introduces GammaTile at the time of initial tumor resection, marking a notable shift from the conventional glioblastoma radiation timeline, which typically delays radiation until several weeks […]
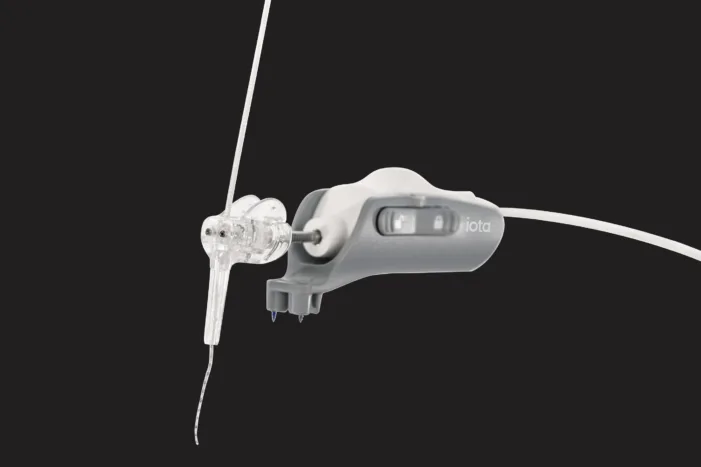
iotaMotion, Inc. has received U.S. Food and Drug Administration 510(k) clearance to expand the approved use of its iotaSOFT Robotic-Assisted Cochlear Implant Insertion System for children as young as four years old. This regulatory milestone makes the iotaSOFT system the first robotic-assisted cochlear insertion platform authorized for use in school-aged pediatric patients, expanding access to […]
Cereno Scientific has announced the peer-reviewed publication of its next-generation HDAC inhibitor CS014 in the Journal of Thrombosis and Haemostasis, revealing the compound’s ability to significantly reduce thrombosis without increasing bleeding risk. The study marks the first formal disclosure of CS014’s chemical structure and nonclinical pharmacology, positioning the compound as a differentiated HDAC inhibitor with […]