Why Cornerstone Robotics’ Sentire CE Mark could shake up Europe’s surgical robotics market
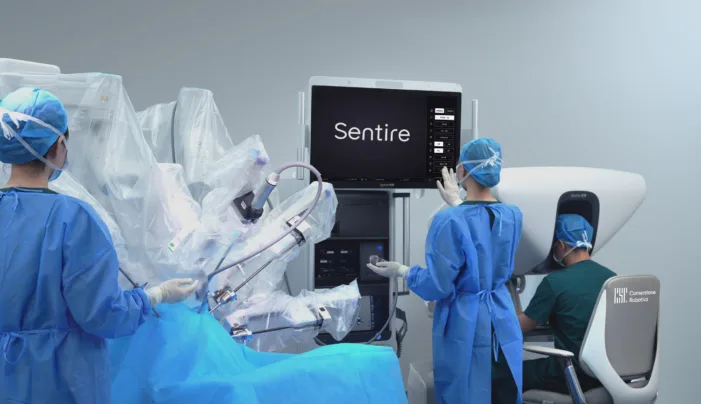
Cornerstone Robotics has cleared Europe’s MDR bar for Sentire. The real test is whether surgical robotics can scale beyond elite hospitals.

Cornerstone Robotics has cleared Europe’s MDR bar for Sentire. The real test is whether surgical robotics can scale beyond elite hospitals.

Find out how Resbiotic and Tiny Health’s at-home microbiome test could reshape consumer gut health, diagnostics and personalised wellness.

Find out how Elmach’s WKH-100 cartoner could improve flexible, GMP-ready pharma packaging for bottles, blisters and tubes.

Find out how Basler’s GMSL vision system could improve lab automation, imaging workflows, and data handling in life sciences.

GHO Capital and CBC Group are creating a $21bn healthcare investor. Find out what it means for pharma, medtech and life sciences funding.

Europe’s STI surge is testing diagnostics, prevention and treatment capacity as gonorrhoea and syphilis reach record levels.

Veracyte’s ASCO data could sharpen the case for genomic testing in breast and prostate cancer. Read why adoption still faces hard tests.
ClearNote Health will present Avantect pancreatic cancer test validation data at ASCO 2026. Find out what it could mean for early detection.
Find out how Hyperfine’s PRIME study could reshape emergency brain imaging with faster bedside portable MRI access.
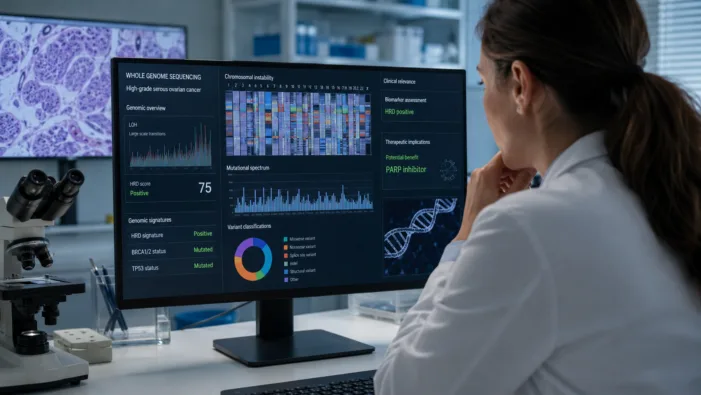
PARP selection still leaves gaps. Inocras’ WGS-HRD data suggest whole-genome testing may refine ovarian cancer maintenance decisions.