Will cartridge-based CBC systems replace central labs? Henry Schein bets on it with CitoCBC

Find out how Henry Schein’s exclusive U.S. deal for CytoChip’s CLIA-waived CitoCBC could reshape point-of-care blood diagnostics.

Find out how Henry Schein’s exclusive U.S. deal for CytoChip’s CLIA-waived CitoCBC could reshape point-of-care blood diagnostics.

EndoQuest wins FDA approval to complete PARADIGM Trial for its ELS system. Find out what this milestone means for robotic GI surgery innovation.

Provation launches iPro Lite for mobile anesthesia documentation. Find out how it could change billing, compliance, and risk in outpatient surgery centers.

iSono Health launches ATUSA™, the first wearable breast ultrasound. Find out how it could transform access, accuracy, and economics in breast imaging.

FDA clears PTOA risk reduction claim for Miach Orthopaedics’ BEAR Implant. Find out what this means for ACL treatment, patients, and future adoption.

VizMark’s VM1 marker receives FDA clearance for artifact-free breast imaging. Find out how this may change tumor localization in dense breast cases.

Amplifi Vascular’s device earns FDA Breakthrough and CMS Category B status. Find out what this means for dialysis access innovation and payer alignment.

Find out how Strive’s acquisition of Semler Scientific strengthens its diagnostic and patient monitoring capabilities in the regulated medical device market.

Find out how AIROS Medical’s five-year warranty on compression devices may shift durability standards and procurement expectations in lymphedema care.
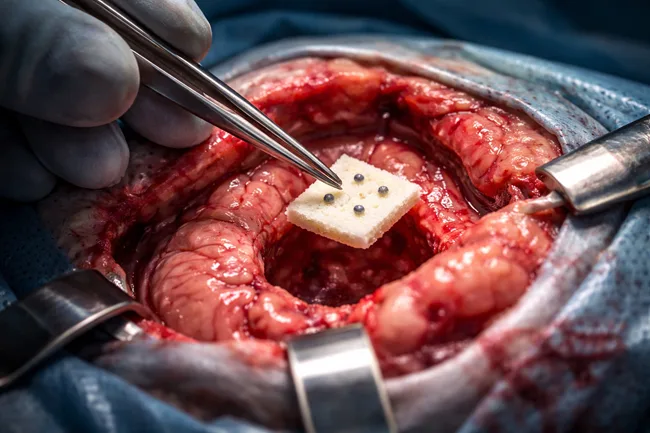
GT Medical Technologies has announced the first patient enrollments in its BRIDGES trial, a randomized clinical study assessing the use of GammaTile therapy in newly diagnosed glioblastoma. The trial introduces GammaTile at the time of initial tumor resection, marking a notable shift from the conventional glioblastoma radiation timeline, which typically delays radiation until several weeks […]