Why BlueWind Medical’s $47.8m raise matters for the next phase of UUI treatment

UUI devices have clinical momentum. BlueWind Medical’s next challenge is proving Revi can scale through access, reimbursement, and workflow adoption.

UUI devices have clinical momentum. BlueWind Medical’s next challenge is proving Revi can scale through access, reimbursement, and workflow adoption.

Cuffs are accurate but inconvenient. Biozen’s FDA-cleared BP1000 tests whether fingertip blood pressure checks can change hypertension care.

Bladder testing needs better real-world data. Bright Uro’s FDA-cleared sensor may push urodynamics beyond catheter-heavy workflows.
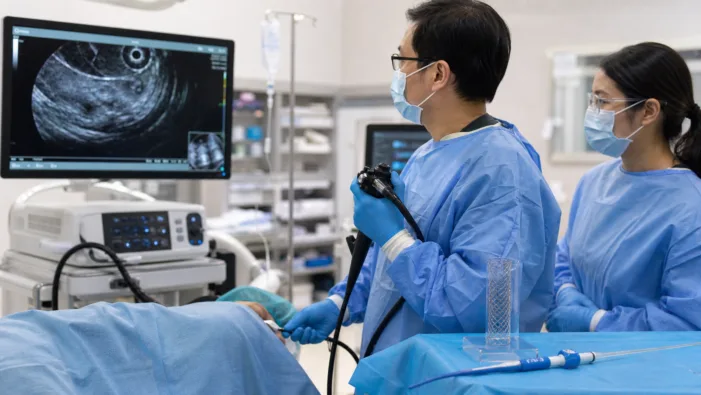
Therapeutic EUS is getting more competitive. TaeWoong Medical USA’s SPAXUS clearance now faces the harder test: real-world adoption.
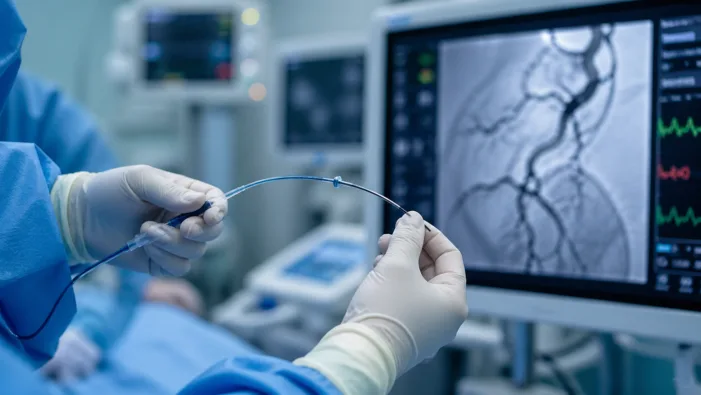
Find out how Sonorous Neurovascular’s BosCATH FDA clearance strengthens its neurovascular access strategy today!

Anumana won FDA clearance for an ECG-AI pulmonary hypertension tool. Read what it changes for diagnosis, workflow adoption, and cardiovascular AI strategy.
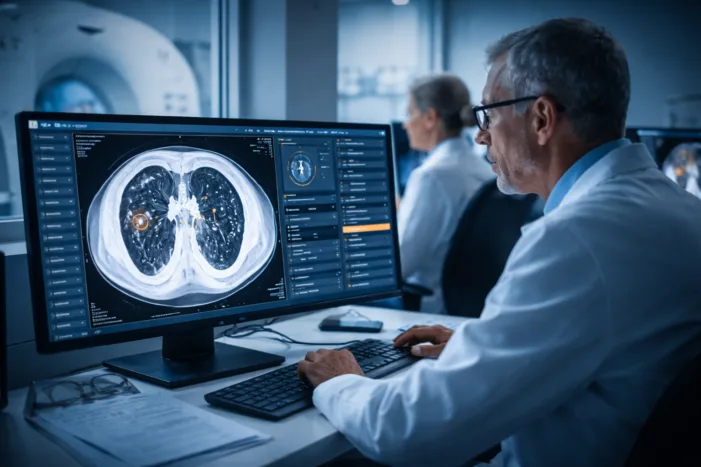
Median Technologies advances eyonis LCS after FDA clearance. Discover what this means for AI lung cancer screening and European expansion.

Dror Ortho-Design secures FDA clearance for ZSmile. Explore what pneumatic aligners mean for orthodontic innovation and adoption.

FDA clears Explant Express for ruptured breast implant removal. Explore what this means for surgical workflows, adoption risks, and explantation standardisation.

KORU Medical’s FreedomEDGE gets FDA clearance for RYSTIGGO delivery in clinics. Find out how this expands access to gMG therapy and reshapes infusion workflows.