Can Chest Wall Innovations turn FDA clearance into adoption in rib fixation?

Rib fixation is gaining clinical ground. Chest Wall Innovations now faces the harder test: converting FDA clearance into surgeon adoption.

Rib fixation is gaining clinical ground. Chest Wall Innovations now faces the harder test: converting FDA clearance into surgeon adoption.
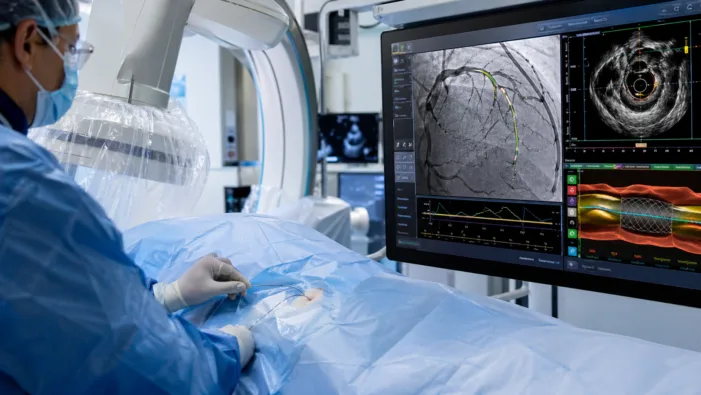
AI can now guide coronary imaging in real time. Abbott’s Ultreon 3.0 puts precision PCI closer to routine clinical workflow.

Boston Scientific owns the PFA market. Pulse Biosciences just posted 90% one-year AF freedom. Is nanosecond PFA a real disruption or an acquisition target?

Utepreva’s FDA-cleared endometrial sampler promises earlier cancer detection. Find out how this device could reshape diagnostics today.
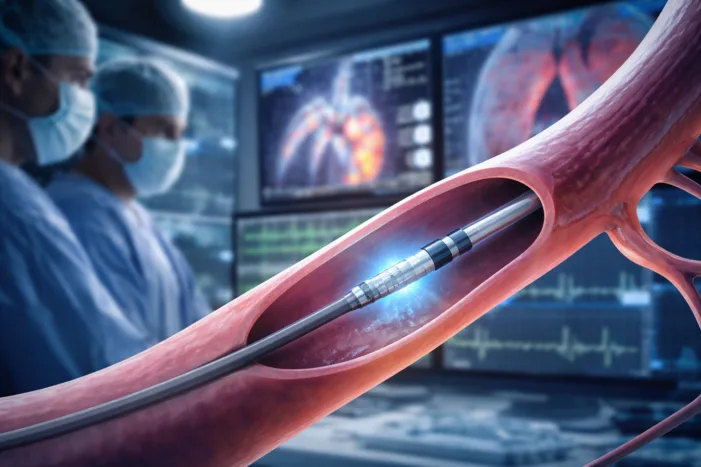
Pulnovo Medical raised $100 million with Medtronic backing. Read what it means for PADN, pulmonary hypertension, heart failure, and device markets.

Miracell has won FDA 510(k) clearance for SMART M-CELL. Read what it changes for PRP, BMAC, adoption, and regenerative medicine competition.

InterVene has completed 30 Recana cases, raising new questions about venous ISR treatment, adoption, and launch strategy. Read the full analysis.
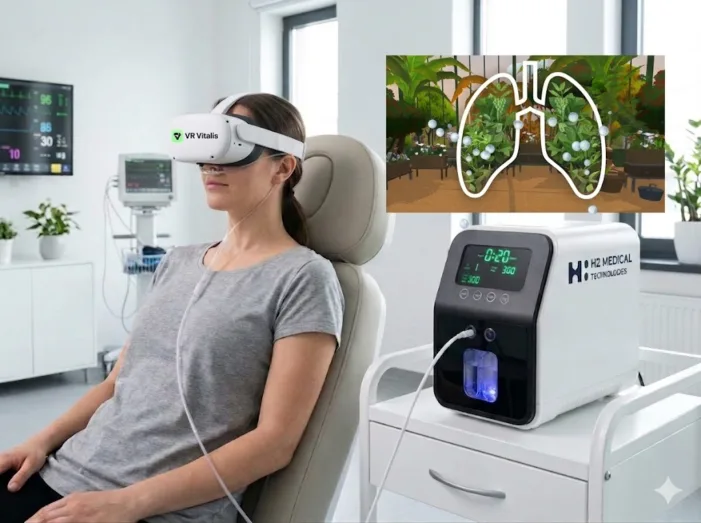
Can VR LIFE and H2 Global Group scale from European validation to global MedTech relevance? Read the deeper analysis now.
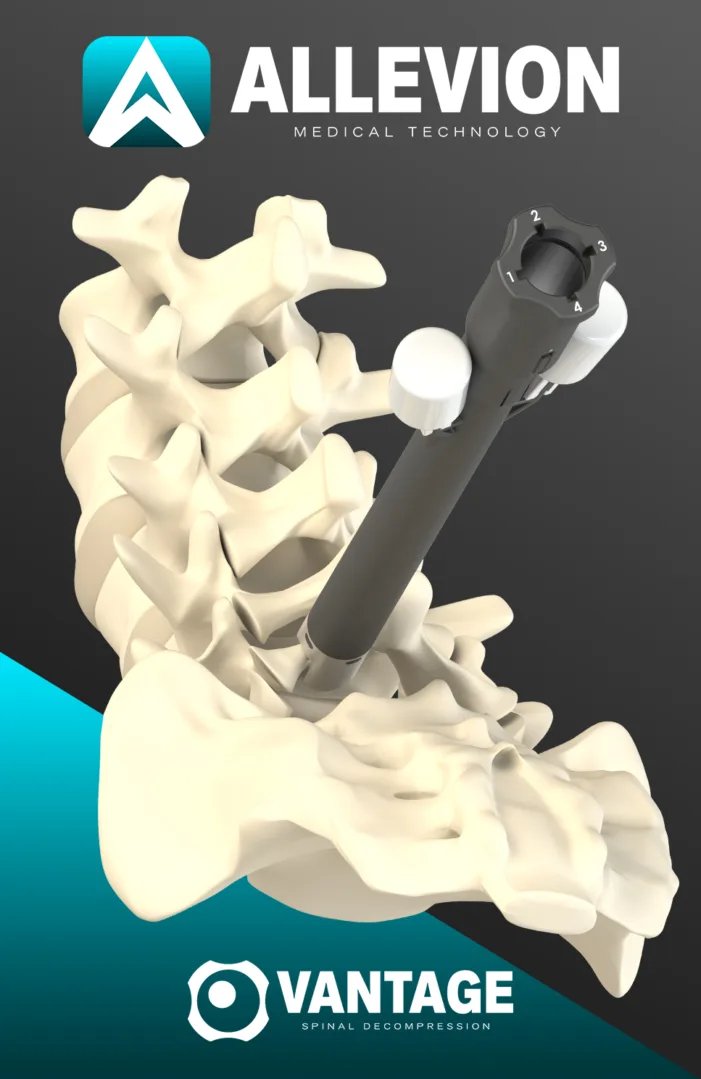
Allevion Medical’s Vantage won FDA 510(k) clearance for lumbar decompression. Read what this may change for spinal stenosis workflow, adoption, and risk.

ATL Medical and OMNIVISION have expanded arthroscopy imaging integration. Read what this means for OEM speed, quality, and device development.