HistoSonics is taking histotripsy beyond liver tumors. Kidney cancer care may be the next credibility test.
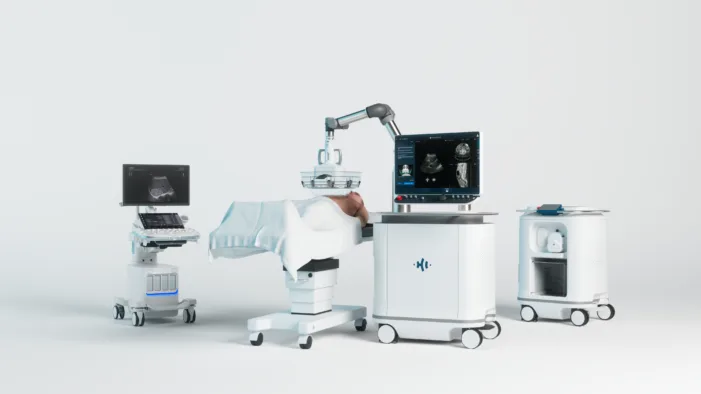
HistoSonics has liver clearance. Kidney tumors are the harder test, where non-invasive ambition must meet regulatory and clinical proof.

HistoSonics has liver clearance. Kidney tumors are the harder test, where non-invasive ambition must meet regulatory and clinical proof.
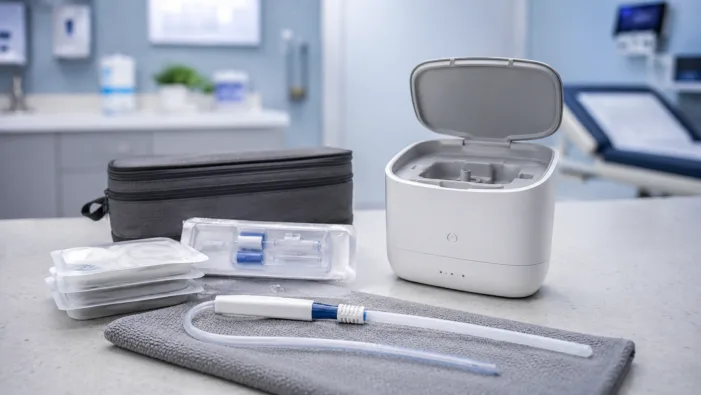
Single-use catheters finally face a regulated reuse challenge. Aurie’s FDA De Novo win could shift urology device economics.

Cuffs are accurate but inconvenient. Biozen’s FDA-cleared BP1000 tests whether fingertip blood pressure checks can change hypertension care.

Could fully implanted cochlear implants transform hearing restoration? Explore the regulatory, clinical, and market implications now.
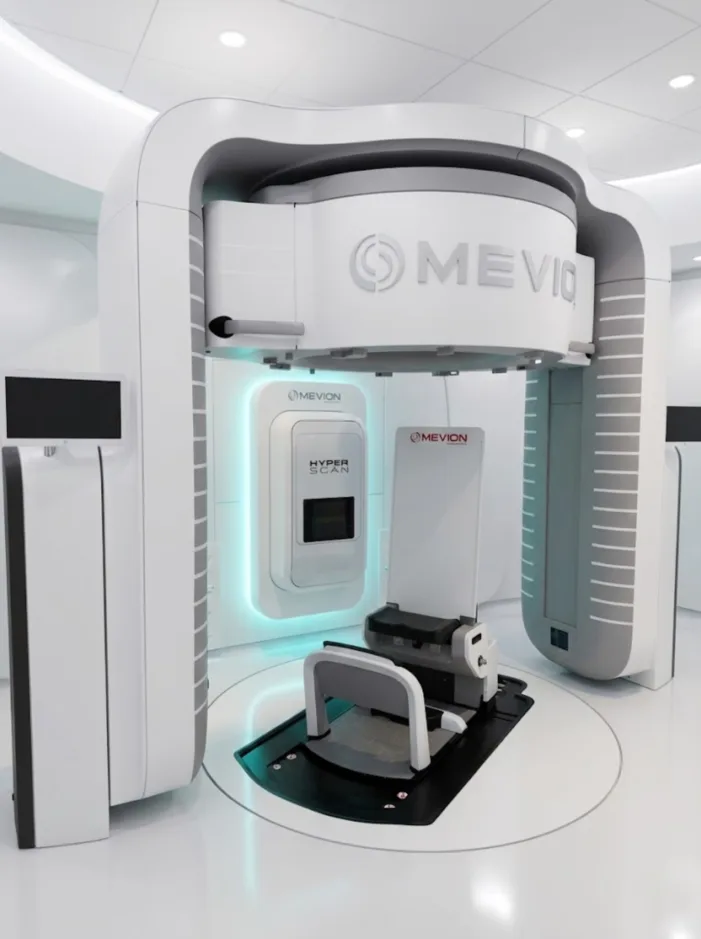
Proton therapy has long had an infrastructure problem. Mevion’s S250-FIT asks whether existing LINAC vaults can change that.

Inhalers are getting smarter, but complexity can backfire. Phillips Medisize is betting DPI success starts with patient-centric design.

Sensus settled the dispute, but the harder test is SRT adoption. Can reimbursement momentum turn into durable medtech growth?

Saudi localization gives Co-Diagnostics a new opening. The test is whether CoMira can turn a factory lease into diagnostics traction.

Hospitals need safer medication delivery, not more disconnected tech. BD and Wellstar are testing whether AI can close that workflow gap.

ECMO transport is a clinical weak point. Medtronic’s VitalFlow CE mark tests whether device mobility can strengthen critical care networks.