What PepGen’s 5 mg/kg FREEDOM2 cohort reveals about the risk-reward profile of PGN-EDODM1

PepGen’s FREEDOM2 data hint at PGN-EDODM1 activity in DM1, but bigger questions remain. Read what the 5 mg/kg cohort really changes.

PepGen’s FREEDOM2 data hint at PGN-EDODM1 activity in DM1, but bigger questions remain. Read what the 5 mg/kg cohort really changes.

Read why Sarepta Therapeutics’ first siRNA data in FSHD1 and DM1 could matter for rare disease drug development and investor sentiment.
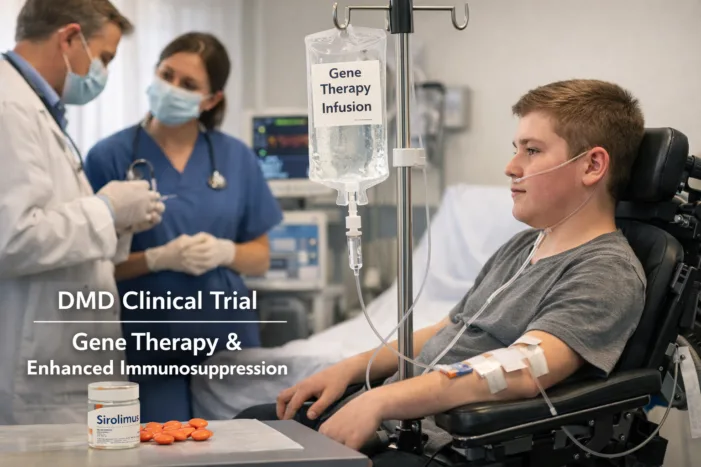
Sarepta’s ELEVIDYS Cohort 8 targets non-ambulatory Duchenne patients with a sirolimus regimen. What the trial design reveals about the path to label restoration.

BridgeBio Pharma reports Phase 3 BBP-418 results and prepares FDA filing. Could this become the first therapy for limb-girdle muscular dystrophy?

NEJM publication of Avidity Biosciences delpacibart etedesiran data raises new questions for DM1 treatment. Discover what this changes next.

Scribe Therapeutics hits a second in vivo CRISPR milestone with Eli Lilly. Explore what this changes for gene editing platforms and future therapies.

Sarepta’s gene therapy ELEVIDYS shows 3-year durability in Duchenne muscular dystrophy. Find out what it means for patients, payers, and global rollout.

Swissmedic approves Santhera’s AGAMREE for Duchenne muscular dystrophy. Find out how vamorolone could reshape long-term steroid treatment strategies.