What Galderma’s nemolizumab phase II data mean for the competitive biologic landscape in pediatric eczema
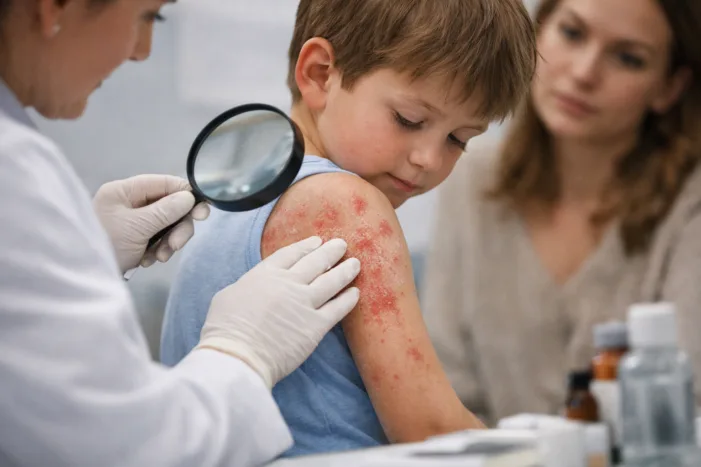
Galderma’s nemolizumab phase II data in children aged 2-11 show 52-week itch and lesion relief. Read the analysis.

Galderma’s nemolizumab phase II data in children aged 2-11 show 52-week itch and lesion relief. Read the analysis.

FDA approves Novartis Cosentyx for adolescent hidradenitis suppurativa. Discover what this decision means for biologic treatment strategies.

Arcutis Biotherapeutics takes full control of ZORYVE promotion. See how this shift could affect real-world dermatology and pediatric prescribing.

Alphyn Biologics has secured $25 million in a twice-oversubscribed Series B financing round to support its clinical pipeline for dermatologic conditions. The funding will enable the company to advance a global Phase 2b trial of its lead asset, Zabalafin Hydrogel, for atopic dermatitis, while also launching a second Phase 2 program for molluscum contagiosum virus. […]