Can MD Hyperbaric turn medical-grade HBOT into a mainstream recovery clinic model?
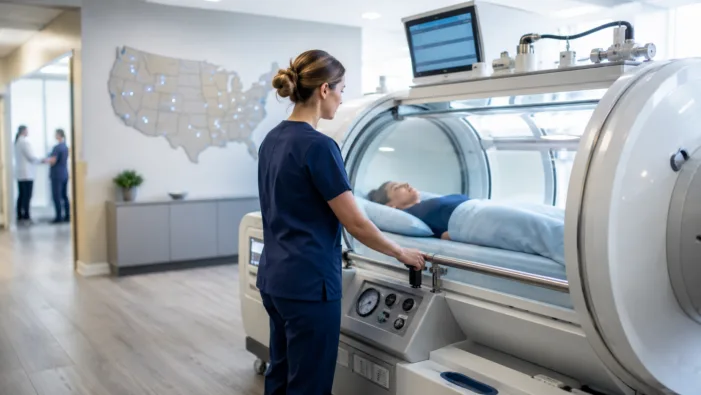
HBOT demand is rising beyond hospitals. MD Hyperbaric’s clinic expansion tests whether recovery care can scale without losing clinical rigor.

HBOT demand is rising beyond hospitals. MD Hyperbaric’s clinic expansion tests whether recovery care can scale without losing clinical rigor.
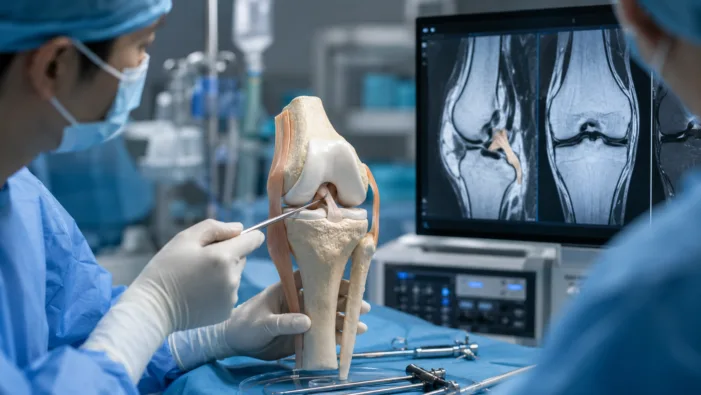
ACL surgery needs better graft choices. CoreTissue BioEngineering’s CT-ACL001 now faces the harder randomized test.

Miracell has won FDA 510(k) clearance for SMART M-CELL. Read what it changes for PRP, BMAC, adoption, and regenerative medicine competition.

FDA clears PTOA risk reduction claim for Miach Orthopaedics’ BEAR Implant. Find out what this means for ACL treatment, patients, and future adoption.