Aesyra SA has announced positive clinical results from its NCT06153810 study evaluating AesyBite Active, a smart biofeedback-enabled nightguard for the treatment of sleep bruxism. The study, which enrolled 26 adult patients, reported a 60.6% reduction in bruxism duration per hour (p < 0.001), surpassing its predefined efficacy threshold. The Swiss-based medtech firm is targeting FDA clearance in 2026 and planning global commercialization thereafter.
Why this biofeedback trial matters for bruxism therapy, oral device adoption, and digital sleep medicine
The successful completion of Aesyra’s clinical trial with AesyBite Active marks a potential turning point for how clinicians and industry observers evaluate sleep bruxism therapies. The study demonstrated not only statistical significance but also clinical meaning in reducing the duration, frequency, and intensity of bruxism episodes. These findings offer validation for a class of interventions that remain underrepresented in mainstream treatment guidelines and often relegated to symptom management.
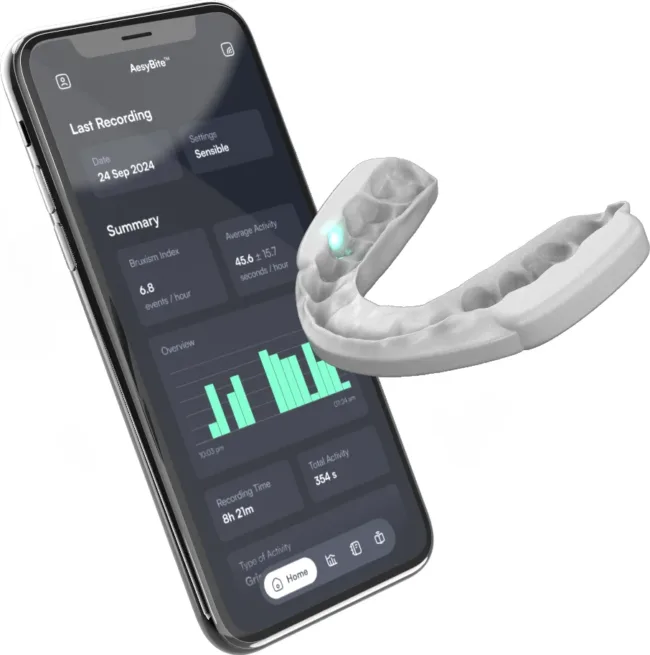
Sleep bruxism remains a poorly understood condition, frequently occurring in patients with no awareness of their nighttime grinding or jaw clenching behavior. Conventional management options typically include passive occlusal splints, which provide a physical barrier but offer no mechanism for reducing bruxism itself. These legacy devices are often associated with limited patient adherence due to discomfort, bulkiness, or perceived ineffectiveness.
Aesyra SA’s AesyBite Active attempts to reframe the category. Rather than protecting the teeth from damage, it seeks to interrupt and diminish the harmful activity in real time using embedded sensors and targeted biofeedback. This active intervention model is gaining traction across other segments of digital health but is still in its early days within oral medicine. The ability to deliver a non-disruptive intervention during sleep—and to do so consistently across severity levels—represents a meaningful step forward in therapeutic design.
The clinical results are especially notable given that the therapeutic effect was observed regardless of baseline bruxism severity or the timing of biofeedback activation. That consistency implies a robustness in performance that could make the device attractive not only to specialists but also to general practitioners and dental sleep medicine providers operating in lower-volume practices.
What Aesyra’s platform reveals about the future of intelligent oral therapeutics
The implications of Aesyra’s technology extend beyond bruxism. By demonstrating real-time therapeutic efficacy in a passive, wearable format, the company is signaling the emergence of a new generation of intelligent oral appliances capable of both monitoring and intervening during sleep.
This evolution mirrors broader shifts in digital therapeutics, where wearables increasingly bridge diagnostic and therapeutic functions. What makes AesyBite Active stand out is its integration of biofeedback into a device designed for long-term, home-based use without requiring patient training or significant behavioral change. The goal is not just efficacy, but scalable, comfortable compliance.
Aesyra’s roadmap, which includes AesyBite Custom—a platform leveraging intraoral 3D scanning and additive manufacturing—further underscores its ambition to build a modular digital dentistry ecosystem. This strategy aligns with a growing trend among medtech innovators to deliver platform-based offerings that address multiple indications or segments through a shared sensor, software, and customization core.
If Aesyra succeeds, its technology could enable expansion into adjacent indications such as temporomandibular joint disorders, clenching-related headaches, or even the early detection of sleep apnea precursors. The modular nature of the device—custom-fit, digitally scanned, and sensor-enabled—offers a flexible chassis for indication-specific algorithms, much like how CPAP machines evolved into adaptive servo-ventilation systems in the sleep apnea market.
Why the study’s endpoints and safety profile raise regulatory confidence ahead of FDA review
From a regulatory standpoint, Aesyra’s approach appears aligned with current expectations for Class II devices seeking clearance via the FDA’s 510(k) pathway. The company’s ability to demonstrate both statistically significant efficacy and an absence of adverse events in a registered clinical investigation (NCT06153810) lends credibility to its filing.
Although the sample size of 26 patients may be considered small, the study design appears sufficiently powered to meet its primary endpoint, with a reported 60.6% reduction in bruxism duration per hour and a narrow 95% confidence interval (55.8%–64.9%). This level of consistency in the primary outcome, paired with multiple significant secondary endpoints—including reductions in episode count, duration, and intensity—strengthens the overall evidentiary package.
Regulatory watchers suggest that the FDA will likely focus on the device’s durability, consistency across nights, and variability in biofeedback sensitivity as part of its risk-benefit analysis. But the lack of reported side effects, along with favorable patient-reported outcomes related to comfort and acceptance, will likely support a low-risk designation under existing device codes.
Notably, Aesyra is planning both U.S. and European regulatory submissions. Its strategy to pursue FDA clearance ahead of CE marking suggests a focus on entering the North American market first, which may be influenced by the growing integration of dental and sleep specialties in the U.S. healthcare system. Providers operating in sleep labs, dental sleep medicine clinics, and integrated care networks are already experimenting with digital health tools, making Aesyra’s smart oral appliance a timely entrant.
What this means for competitors in oral devices and digital health wearables
The competitive implications of Aesyra’s success are twofold. First, within the traditional dental device market, few companies offer active interventions for bruxism. Most rely on passive splinting technologies, which are typically commoditized and subject to price pressure. If AesyBite Active gains traction, it could begin displacing these lower-margin products in select patient populations, particularly those with moderate to severe bruxism.
Second, for digital health companies and wearable device manufacturers, Aesyra’s work raises the bar for what embedded therapeutics can look like in small-form-factor applications. Most current biofeedback devices for behavior modification are wrist-based or auditory. Translating this capability into a dental appliance that users wear during sleep—without triggering arousals or sleep fragmentation—is a technically challenging feat. It also opens doors for cross-disciplinary collaboration between oral health, neurology, and behavioral sleep medicine.
Clinicians tracking the field are likely to scrutinize longer-term outcomes and real-world usage patterns. While initial comfort and efficacy data are promising, questions remain about device durability, cleaning protocols, and patient adherence over time. Similarly, payers will be looking for cost-effectiveness data that link bruxism suppression with reduced dental procedures, TMJ-related interventions, and improved quality of life.
If Aesyra can successfully position AesyBite Active within existing reimbursement frameworks—either through dental insurance or under emerging DME coding structures—it may unlock broader access and commercial adoption. However, this will likely depend on follow-up studies that capture long-term outcomes, cost offsets, and provider feedback in diverse care settings.