Insulet Corporation, the Massachusetts-based medical device manufacturer behind the Omnipod tubeless insulin pump platform, has released feasibility study results showing its fully closed-loop automated insulin delivery system achieved 68% time in range in adults with type 2 diabetes without requiring any mealtime bolus inputs. The EVOLUTION 2 findings, presented at the Advanced Technologies and Treatments for Diabetes conference in Barcelona, represent the second iteration of a staged feasibility programme underpinning a planned pivotal trial and eventual 510(k) regulatory submission in the United States.
Why a bolus-free result at 68% TIR changes the clinical calculus for type 2 insulin users
The headline metric from EVOLUTION 2 is clinically significant not because 68% time in range is exceptional in absolute terms, but because it was achieved without any user mealtime interaction whatsoever. In automated insulin delivery for type 1 diabetes, hybrid closed-loop systems routinely require patients to announce meals and enter carbohydrate estimates, a burden that directly affects adherence and outcomes. Removing that interaction entirely for a type 2 population, which typically includes older patients, those with lower digital health literacy, and individuals managing multiple comorbidities alongside diabetes, is the design challenge that has eluded the field. The EVOLUTION 2 algorithm cleared that bar in a cohort that included both basal-bolus and basal-only insulin users, as well as individuals transitioning from multiple daily injections to pump therapy, making the participant mix more representatively complex than many early feasibility studies.
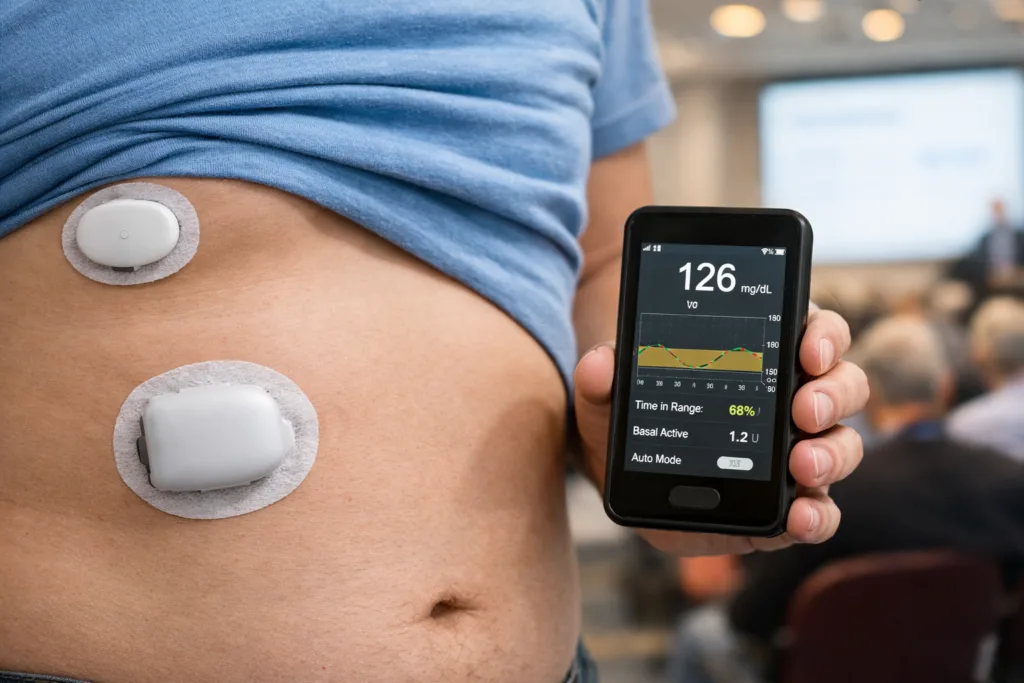
The improvement from a baseline of approximately 44% time in range to 68% represents roughly three and a half additional hours per day within target glucose limits. Against the backdrop of population-level data showing that only around one quarter of adults with type 2 diabetes on insulin currently achieve an HbA1c at or below 7.0%, the magnitude of that shift is commercially and clinically meaningful. Critically, the improvement came without increasing hypoglycaemia: time below range held at a median of 0.14% below 70 mg/dL, well under the American Diabetes Association threshold of 4%, and no severe hypoglycaemic or diabetic ketoacidosis events were reported across the study.
What EVOLUTION 2 reveals about algorithm maturity and the gap to pivotal-grade evidence
Insulet’s decision to run a multi-version algorithm evaluation within a single feasibility study reflects a deliberate accelerated development model rather than the conventional sequential study approach. Investigators tested multiple algorithm iterations within the same participant cohort, allowing rapid identification of the highest-performing configuration. That design choice compresses the development timeline but limits certain types of cross-study comparability and makes it more difficult to draw definitive conclusions about which specific algorithmic changes drove the improvement between EVOLUTION 1 and EVOLUTION 2. EVOLUTION 1, published in late 2024, reported a time in range increase from 52% to 65% without boluses. The move to 68% in EVOLUTION 2 reflects continued refinement, but the incremental gain between the two studies is modest enough that the pivotal EVOLVE trial, planned for 2026, will need to demonstrate both the magnitude and durability of benefit in a larger, adequately powered population.
The EVOLUTION 2 cohort included 24 adults from New Zealand, aged 16 to 70, with an average diabetes duration of 16 years. That is a small and geographically homogeneous sample for a feasibility study, which the investigators themselves positioned as a platform for iterative algorithm development rather than as evidence of efficacy. The high continuation rate into the extension phase, above 90% of participants, is a useful signal of tolerability and user experience, though continuation willingness in a closely monitored clinical setting should not be read as a direct proxy for real-world adherence in a commercial launch scenario. Diversity within the cohort across sex, ethnicity, baseline HbA1c, and body mass index is methodologically important but the absolute numbers involved mean subgroup analyses remain exploratory.
How Insulet’s type 2 positioning differs from the existing AID competitive landscape
The automated insulin delivery market is currently dominated by hybrid closed-loop systems approved primarily for type 1 diabetes, including Insulet’s own Omnipod 5, Medtronic’s MiniMed systems, and Tandem Diabetes Care’s Control-IQ platform. None of these systems are indicated for type 2 diabetes management at the population level, and none operate in a fully closed-loop configuration that eliminates all mealtime user actions. Insulet’s development programme therefore targets a largely unaddressed segment rather than competing head-to-head with existing cleared devices. The strategic rationale is straightforward: the type 2 diabetes population on insulin is substantially larger than the type 1 population, tends to be managed in primary care rather than specialist diabetes centres, and has historically been poorly served by device-based therapies because of their complexity and cost.
The competitive risk for Insulet is not primarily from other pump manufacturers in the near term, but from the rapid expansion of GLP-1 receptor agonist therapy and dual or triple agonist agents that are reshaping the treatment algorithm for type 2 diabetes. As these agents produce substantial glycaemic and weight-related benefits with once-weekly dosing and no hypoglycaemia risk, the addressable population for insulin-based therapy may contract over the next decade. Industry observers tracking the diabetes device space note that the clearest near-term market for a fully closed-loop type 2 system may be patients who are already on insulin and cannot transition to GLP-1-based regimens due to renal impairment, cost barriers, tolerability issues, or insufficient glycaemic response.
Regulatory pathway and commercial timeline: what the 2028 target requires
Insulet has publicly outlined a timeline that moves from the EVOLVE pivotal study in 2026 to a 510(k) submission in 2027 and a commercial launch in 2028. That schedule is ambitious but not implausible for a 510(k) pathway, which is the appropriate regulatory route given the use of predicate devices within the automated insulin delivery category. However, the FDA’s De Novo and 510(k) pathways for novel AID configurations have involved iterative feedback cycles in previous submissions, and a first-in-class fully closed-loop indication for type 2 diabetes may face additional scrutiny around the definition of the intended use population, the adequacy of hypoglycaemia safeguards for a population with variable insulin sensitivity, and the robustness of real-world usability data.
Regulatory watchers note that the current investigational device status of the Insulet FCL system means no comparative efficacy data against other insulin delivery methods, beyond standard injection therapy, has yet been generated in a registration-grade study. The EVOLVE pivotal design, endpoints, and comparator arm structure will be closely scrutinised by clinicians and payers when they become available. Reimbursement in the United States for CGM-integrated AID systems has expanded significantly in recent years, but coverage frameworks for type 2 diabetes remain more restrictive than for type 1, and a new product category targeting type 2 patients on insulin will need to build a health economics case that goes beyond time in range metrics to include hospitalisations, hypoglycaemia-related costs, and quality-of-life outcomes.
Risks and unresolved questions heading into the pivotal phase
Several material uncertainties remain ahead of the pivotal study. The fully closed-loop algorithm’s performance in real-world conditions, including irregular sleep patterns, physical activity, illness, and concomitant medications that affect insulin sensitivity, has not been assessed outside a controlled feasibility context. Type 2 diabetes is a heterogeneous condition with substantial variation in residual beta cell function, insulin resistance, and comorbidity burden, and algorithm generalisation across that spectrum will require a larger and more diverse population than either EVOLUTION study has enrolled.
The durability of glycaemic improvement beyond the study period is also unestablished. Insulet reported that more than 90% of EVOLUTION 2 participants elected to continue into an extension phase, which provides ongoing data collection opportunity, but the commercial analogue of that engagement level under real-world conditions, without the motivational effects of clinical trial participation, will only be visible after launch. The device and algorithm interaction with high-dose basal insulin requirements common in longer-duration type 2 diabetes patients, as well as the system’s behaviour during intercurrent illness or surgical episodes, represent further areas where clinicians will want to see data before broad adoption. Insulet’s EVOLUTION programme has delivered encouraging proof-of-concept findings; what the field now needs is the size, duration, and design rigour of a pivotal study to determine whether fully closed-loop delivery can genuinely redefine outcomes for the large and underserved population living with type 2 diabetes on insulin.