Everads Therapy has published first-in-human clinical data for the Everads Injector in Ophthalmology Science, reporting safety, tolerability, procedural feasibility, and device performance in suprachoroidal administration of a triamcinolone acetonide suspension for diabetic macular edema. The clinical-stage ophthalmology technology developer also disclosed three ARVO 2026 presentations focused on infrared thermography, injection pattern confirmation, and standardized assessment of suprachoroidal drug delivery.
Why the Everads Injector data matters for the future of retinal drug delivery
The significance of the Everads Therapy update is not only that the Everads Injector has reached first-in-human publication. The more important issue is whether the device can help make suprachoroidal delivery more predictable, scalable, and clinically usable in routine retina practice. For a field already familiar with repeated intravitreal injections, any new delivery route must clear a high bar. It must not simply be clever. It must be reliable, reproducible, acceptable to clinicians, and capable of supporting drugs whose value depends on reaching the posterior segment of the eye efficiently.
Suprachoroidal delivery has long attracted interest because it offers a route between conventional intravitreal injection and more invasive surgical approaches. The suprachoroidal space sits between the sclera and choroid, creating a potential channel for targeted distribution toward the retina, choroid, macula, and posterior pole. In theory, that route could support localized delivery while limiting exposure to structures that do not need the drug. In practice, however, the challenge has always been controlled access. The eye is a small, unforgiving structure, and drug delivery innovation in ophthalmology tends to advance only when clinicians believe the technique can be performed consistently without adding procedural burden.
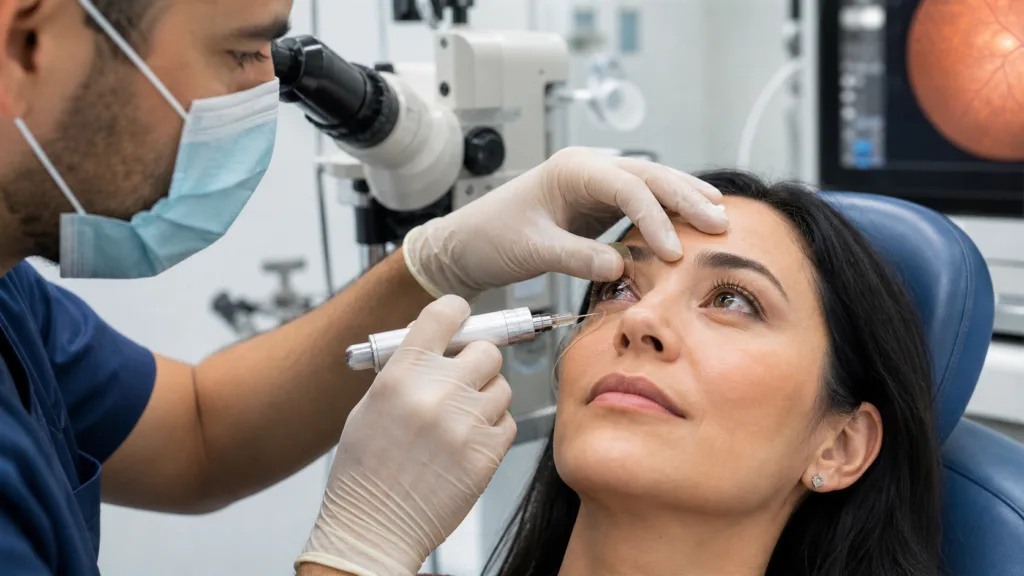
This is where Everads Therapy is trying to differentiate the Everads Injector. The medical device platform uses a non-sharp tissue separator to access the suprachoroidal space through controlled tissue separation, followed by tangential delivery toward the posterior segment. That design choice matters because it frames Everads Therapy’s approach less as a simple injector and more as a delivery system intended to reduce uncertainty around access, directionality, and flow. The publication’s finding of rapid, posteriorly directed drug distribution, visualized through thermal imaging, is therefore the key signal. It suggests that the device may not only place material into the suprachoroidal space, but may help direct it toward the region most relevant for retinal disease.
What thermal imaging adds to the evidence package beyond procedural feasibility
The use of thermal imaging gives the Everads Therapy data a layer of interest beyond conventional early device safety reporting. Many first-in-human device studies can show whether a procedure was completed and whether immediate safety concerns emerged. Fewer are able to visualize, in real time, how delivery behaves once administration occurs. For a technology built around spatial distribution, that distinction matters.
Infrared ocular thermography could become a useful evidentiary tool if it helps clinicians and developers distinguish between successful suprachoroidal delivery, intravitreal delivery, and other injection patterns. Everads Therapy’s ARVO 2026 presentations appear to build directly on that question, including work on real-time thermography for confirming suprachoroidal delivery in humans and differentiating intravitreal from suprachoroidal injection patterns. That points to a broader industry need: as new delivery routes emerge, the sector needs better ways to prove not only that a drug was administered, but that it went where the developer says it went.
The limitation is that imaging confirmation does not automatically establish long-term clinical benefit. Thermal imaging may strengthen confidence in device mechanics, but it does not answer whether a drug delivered by this route produces superior durability, improved anatomical outcomes, better visual acuity, fewer adverse events, or lower treatment burden compared with established delivery methods. The Everads Therapy study used triamcinolone acetonide suspension in diabetic macular edema, a logical early testing context because corticosteroid delivery has an established role in retinal disease. However, the broader commercial and clinical case will depend on whether the platform can support a range of therapeutic modalities, including small molecules, biologics, cell therapies, and gene therapies, without compromising safety or consistency.
How the platform compares with existing retinal treatment delivery models
Retina specialists are already operating within a heavily procedure-based treatment model. Anti-VEGF injections have transformed the management of retinal vascular disease, but they also created a recurring burden for patients, clinics, caregivers, and payers. Intravitreal injection is familiar, widely adopted, and relatively efficient in experienced hands. That familiarity is both the benchmark and the barrier for suprachoroidal systems such as the Everads Injector.
For Everads Therapy, the opportunity lies in diseases and therapeutic classes where conventional delivery is imperfect. If suprachoroidal delivery can concentrate drug exposure closer to the posterior tissues of interest, it could be attractive for therapies that need targeted distribution, sustained activity, or reduced off-target exposure. The company’s stated interest in gene therapy, cell therapy, small molecule formulations, and other modalities reflects that broader ambition. In ophthalmology, delivery platforms can become strategically powerful when they help unlock therapies that otherwise face anatomical, tolerability, or dosing challenges.
The risk is that retina specialists may not adopt a new route unless the clinical advantage is obvious. Office-based administration helps, because it preserves the workflow logic of modern retina practice. Still, ease of use must be proven across operators, patient anatomies, disease states, and real-world clinic conditions. A device that performs well in a controlled early study may face tougher questions when deployed across high-volume settings. Clinicians will want to know how steep the learning curve is, how often delivery fails or requires repositioning, whether patient discomfort differs from intravitreal injection, and whether adverse events remain manageable as exposure expands.
Why diabetic macular edema is a useful but limited proving ground
Diabetic macular edema is a clinically relevant early setting for testing the Everads Injector because it involves posterior segment pathology, chronic treatment demand, and a patient population where durability and treatment burden matter. A triamcinolone acetonide suspension also gives researchers a practical way to evaluate suprachoroidal corticosteroid delivery without immediately moving into more complex therapeutic categories. From an early development standpoint, that makes sense.
However, diabetic macular edema is not a complete proxy for the full commercial promise of Everads Therapy’s platform. The most valuable future applications may involve therapies where delivery itself is a major constraint. Gene therapy is an obvious example because retinal targeting, dose control, immune response, durability, and procedural complexity all influence product design. Cell therapy and other advanced modalities could create similar delivery challenges. If Everads Therapy wants to position the Everads Injector as a broad enabling platform, the next evidence layers will need to move beyond basic feasibility and toward modality-specific performance.
That creates a strategic fork. Everads Therapy can either continue generating device-centric evidence across multiple contexts, or it can deepen the clinical case around a narrower set of high-value applications. The former supports partnership discussions by showing versatility. The latter may produce stronger proof for regulators, clinicians, and potential commercial partners. The better path may depend on whether Everads Therapy prioritizes platform licensing, co-development with drug developers, or eventual product-specific regulatory strategies.
What regulators and industry partners are likely to watch after ARVO 2026
Regulatory watchers are likely to focus on how Everads Therapy defines the Everads Injector’s role in future clinical programs. If the device is used to deliver already approved or well-characterized agents, the regulatory questions may center on device performance, procedural safety, and route-specific risks. If it becomes part of novel drug-device combinations, including advanced therapies, the path may become more complex. In ophthalmology, combination development can require close alignment between therapeutic effect, delivery reliability, manufacturing control, and user training.
Industry partners will also look for reproducibility. A single first-in-human publication is useful, but partnership confidence usually comes from repeated evidence across settings. The ARVO 2026 presentations could help by showing whether Everads Therapy can build a coherent evidence package around imaging, flow pattern differentiation, and standardized assessment. That matters because drug developers evaluating delivery platforms do not only ask whether the device works once. They ask whether it can become part of a development program without creating new regulatory, clinical, or manufacturing uncertainty.
There is also a reimbursement and workflow question hiding underneath the clinical excitement. Office-based delivery is attractive because it fits current retina infrastructure, but reimbursement frameworks may need to reflect the device, procedure, drug, and follow-up burden. If the platform improves outcomes or reduces injection frequency, the value argument strengthens. If it simply adds a new procedural option without clear clinical or economic differentiation, adoption may be slower.
Why Everads Therapy’s real test begins after first-in-human validation
The Everads Therapy update should be seen as an important early validation step, not as proof that suprachoroidal delivery will displace existing retinal treatment pathways. The favorable safety, tolerability, feasibility, and posterior distribution findings give the Everads Injector a credible starting point. The use of thermal imaging also adds a technically interesting layer, especially because delivery confirmation could become increasingly important as ophthalmology moves toward more sophisticated drug-device combinations.
The harder test begins now. Everads Therapy needs to show that controlled suprachoroidal access can translate into outcomes that clinicians, regulators, payers, and partners can value. In practical terms, that means larger studies, clearer comparative evidence, stronger procedural reproducibility, and a tighter link between distribution patterns and clinical benefit. For now, the Everads Injector appears to be moving the conversation from whether suprachoroidal access is technically possible toward whether it can become a scalable platform for retinal therapy development.
That shift matters. Ophthalmology innovation is no longer only about better molecules. It is increasingly about whether the right therapy can reach the right tissue, in the right concentration, with acceptable safety and clinic-level efficiency. Everads Therapy has placed itself inside that larger delivery debate. The next phase will show whether the Everads Injector remains an intriguing device story or becomes part of the infrastructure for the next generation of retinal disease treatment.