GE HealthCare’s Photonova Spectra photon-counting computed tomography system has received 510(k) clearance from the U.S. Food and Drug Administration, giving the medical technology company regulatory permission to bring its newly unveiled spectral photon-counting platform to the U.S. market. The clearance comes shortly after the system’s debut at the Radiological Society of North America 2025 annual meeting and positions Photonova Spectra as GE HealthCare’s latest push into high-end computed tomography imaging, backed by its Deep Silicon detector architecture, 8-bin energy resolution, and one-scan universal workflow.
What matters now is not simply that GE HealthCare has added another computed tomography scanner to its portfolio, but that photon-counting technology is increasingly becoming the next major competitive battleground in advanced diagnostic imaging. Conventional computed tomography systems have already gone through years of optimisation around speed, reconstruction, and dose efficiency. Photon-counting computed tomography offers something more structurally disruptive because it changes how X-ray information is captured at the detector level. By directly counting individual photons and measuring their energy, instead of converting X-rays into visible light first, the technology promises sharper spatial resolution, better spectral separation, and richer tissue characterisation. In practical market terms, that means vendors are no longer selling only image quality improvements. They are increasingly selling a new layer of diagnostic information density.
Why GE HealthCare’s photon-counting computed tomography clearance matters beyond another scanner launch
For GE HealthCare, the clearance has significance well beyond a routine product approval. Photon-counting computed tomography has become a prestige category in imaging because it sits at the intersection of clinical differentiation, premium capital equipment pricing, and future software monetisation. Hospitals do not buy these systems only for incremental image improvements. They buy them for strategic reasons, including referral differentiation, oncology imaging precision, cardiac imaging expansion, and access to advanced quantitative tools that can support research programmes and high-acuity service lines. That makes this category unusually important for both market share and brand leadership.
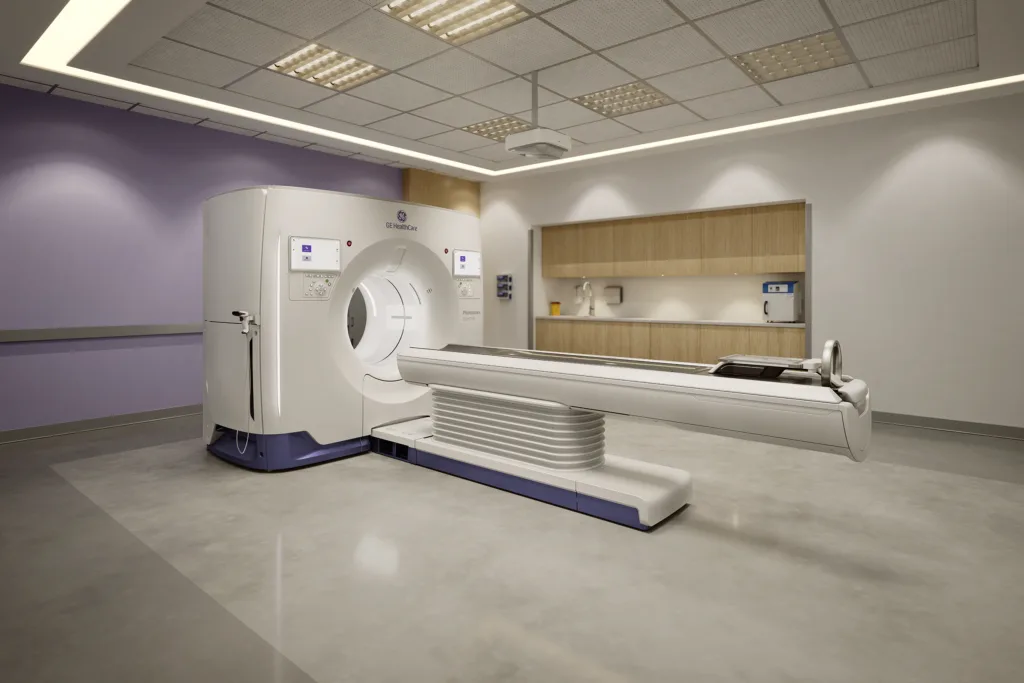
The timing also matters. GE HealthCare is framing Photonova Spectra as part of a broader innovation cycle tied to its multibillion-dollar product investment strategy, with management expecting the wider innovation wave to contribute to revenue growth. That makes Photonova Spectra not just a technology announcement but part of a larger financial narrative. In other words, the U.S.-based imaging manufacturer is using this platform to signal that its research spending is beginning to translate into commercially relevant hardware that can move the revenue needle, especially in premium imaging segments where replacement cycles are slower but margins and prestige are higher.
What Deep Silicon and 8-bin energy resolution could change in clinical imaging workflows
The genuinely new element in GE HealthCare’s pitch is its emphasis on Deep Silicon detector technology and 8-bin energy resolution. In the photon-counting category, the detector is not a background technical detail. It is the entire thesis. The industry has long understood that detector design shapes spectral fidelity, count rate capability, noise behaviour, and the ability to distinguish between materials. GE HealthCare is positioning silicon not merely as an alternative material but as a route to cleaner spectral performance and more precise energy measurement. If that claim holds in routine clinical settings, the detector architecture could become the real competitive moat rather than the scanner branding around it.
The 8-bin energy resolution claim is especially notable because it suggests more refined material discrimination than many clinicians associate with earlier spectral workflows. In theory, better energy discrimination could improve the separation of iodine, calcium, and fat, while also supporting more advanced tissue characterisation in oncology, vascular imaging, and complex abdominal studies. That is where photon-counting computed tomography begins to shift from being a premium image-quality story to becoming a decision-support story. When a scan can provide more usable diagnostic contrast from one acquisition, the technology starts influencing protocol design, follow-up decisions, and potentially the number of additional studies a patient may need.
Yet the caution here is obvious. Better energy separation on paper does not automatically translate into better clinical decisions at scale. The field still has to prove that richer spectral information will be used consistently across busy imaging departments rather than confined to specialist centres. Many technically impressive imaging capabilities struggle to achieve routine adoption because the workflow burden lands on radiologists and technologists who are already operating under productivity pressure. The long-term test will be whether Photonova Spectra’s spectral richness reduces uncertainty without increasing interpretation friction.
Why one-scan spectral workflow claims could matter as much as the detector itself
One of the most commercially important details in the announcement is the one-scan universal workflow. This may sound less glamorous than detector design, but in actual hospital procurement decisions it can matter just as much. Advanced imaging tools often fail to reach their full potential because they require protocol complexity, exam-specific setup, or reconstruction decisions that are difficult to standardise across operators and sites. GE HealthCare is clearly trying to remove that barrier by presenting Photonova Spectra as a system that captures both spectral and ultra-high-definition spatial data automatically in every exam.
If that workflow performs as advertised, the system could lower one of the biggest adoption hurdles in spectral imaging: selective use. Departments often underutilise advanced modes when staff must choose them in advance or adjust protocols for each use case. A universal acquisition model changes the value proposition because it makes spectral data available retrospectively or on demand, not just when someone predicts the need before the scan starts. That could expand the practical utility of the technology in emergency imaging, oncology follow-up, thoracic workups, and cardiac cases where the clinical question evolves after the scan is performed.
Still, universal workflow claims should be treated carefully. Automatic acquisition does not eliminate downstream burdens such as image storage, reconstruction management, reading time, training, and reimbursement ambiguity. Richer datasets are only beneficial if the imaging department can operationalise them without creating bottlenecks. This is where GE HealthCare’s software environment, automation tools, and reconstruction speed become central to the commercial case.
How NVIDIA-backed reconstruction highlights the data burden photon-counting systems must solve
Another revealing element in the announcement is the explicit use of NVIDIA accelerated computing to process what GE HealthCare says can be up to 50 times more data than conventional computed tomography. That point deserves more attention than it may initially receive because photon-counting computed tomography is not just a detector story. It is also a compute story. High-fidelity spectral imaging generates enormous reconstruction demands, and those demands can undermine the clinical value of the platform if image generation becomes slow, inconsistent, or too resource-intensive for routine practice.
By highlighting GPU-powered architecture and CUDA-optimised reconstruction, GE HealthCare is effectively saying it understands that next-generation imaging platforms must be designed as end-to-end systems. The scanner, detector, reconstruction pipeline, and operator workflow must all work together. That matters because premium imaging equipment increasingly depends on integrated hardware-software stacks rather than standalone device performance. In strategic terms, this means the future competitive landscape in computed tomography may resemble other high-performance medical technology categories where compute infrastructure, algorithmic performance, and workflow integration are as important as the underlying hardware.
However, this also introduces fresh questions. Advanced compute infrastructure can improve performance, but it can also increase system complexity, service requirements, and dependence on specific technical ecosystems. Hospitals will want proof that the scanner’s data intensity does not become an operational headache. They will also watch whether advanced processing translates into measurable clinical throughput rather than just impressive engineering specifications.
What clinicians, regulators, and imaging buyers are likely to watch after U.S. clearance
The next phase for Photonova Spectra will depend less on the clearance itself and more on what evidence emerges once the system enters commercial use. Regulatory clearance confirms that the platform can be marketed, but it does not resolve the most important market questions. Clinicians will want to know where photon-counting computed tomography delivers clear superiority over high-end conventional and dual-energy systems, and where the gains are more incremental than transformative. Imaging leaders will look for evidence in oncology lesion detection, coronary imaging, neurological detail, musculoskeletal microstructure, and difficult soft-tissue characterisation, because those are the areas where premium systems must justify their cost.
Academic and translational researchers are also likely to focus on the quantitative imaging opportunities mentioned in the source material, including spectral biomarker discovery and tissue characterisation. That matters because early clinical prestige for these systems is often built through major research institutions that generate publications, protocol innovations, and usage credibility. GE HealthCare’s collaboration with sites such as University of Wisconsin and Stanford Medicine reflects that reality. These institutions are not just evaluation partners. They are credibility engines that can shape how the wider imaging field perceives the platform’s scientific value.
From a commercial standpoint, buyers will watch installation friction, room compatibility, training requirements, service support, and reimbursement logic. GE HealthCare’s claim that Photonova Spectra is designed to fit existing computed tomography ready rooms with minimal changes is strategically important because infrastructure disruption can slow premium equipment adoption. If hospitals can upgrade without major room redesign, the sales conversation becomes easier. Even so, capital budgets remain tight across many health systems, and a technology being clinically impressive does not guarantee immediate purchasing momentum.
Why Photonova Spectra looks strategically important even if broad adoption takes time
The biggest reason this clearance matters is that it reinforces photon-counting computed tomography as a platform transition rather than a niche experiment. GE HealthCare is using Photonova Spectra to show that advanced computed tomography is moving toward richer spectral intelligence, higher material discrimination, and more automated acquisition models. That aligns with a broader industry trend in which scanners are judged not just by speed or dose, but by how much diagnostically useful information they can extract from a single patient encounter.
Even so, the road from clearance to mainstream impact is rarely linear. The unresolved issues are familiar: whether radiologists will fully use the added data, whether hospitals will pay a premium at scale, whether reimbursement will keep pace with capability, and whether performance gains will be obvious enough across diverse patient populations and clinical settings. Photonova Spectra may well prove to be a strong technological step forward, but the real commercial verdict will come from utilisation patterns, not launch messaging.
For now, GE HealthCare appears to have cleared an important hurdle at the right moment. The company has moved quickly from product unveiling to FDA validation, attached the platform to a broader innovation-growth narrative, and built its case around both detector novelty and workflow simplification. That combination gives Photonova Spectra a stronger launch posture than a conventional premium scanner rollout. Whether it becomes a true category-defining system will depend on something much harder than regulatory clearance: proving that better physics can become better routine medicine.