Kallisio has begun clinical implementation of its FDA-cleared Stentra system at Stanford Health Care, marking the first reported integration of the patient-specific intraoral device into a major U.S. hospital’s workflow for head and neck cancer radiation therapy. The system, which received 510(k) clearance in January 2024, enables individualized immobilization of oral structures during intensity-modulated radiation therapy (IMRT), aiming to improve geometric accuracy while protecting healthy tissue.
What this integration reveals about real-world readiness for intraoral positioning tools
Stentra’s adoption by Stanford Health Care is more than a routine post-clearance rollout. It signals the first major proof point that precision intraoral stenting can be operationalized at scale within high-throughput radiation oncology departments. Despite the availability of multiple immobilization techniques for head and neck cancer—including thermoplastic masks, bite blocks, and vacuum cushions—few offer the same level of anatomical fidelity and patient-specific geometry as a digitally designed oral stent.
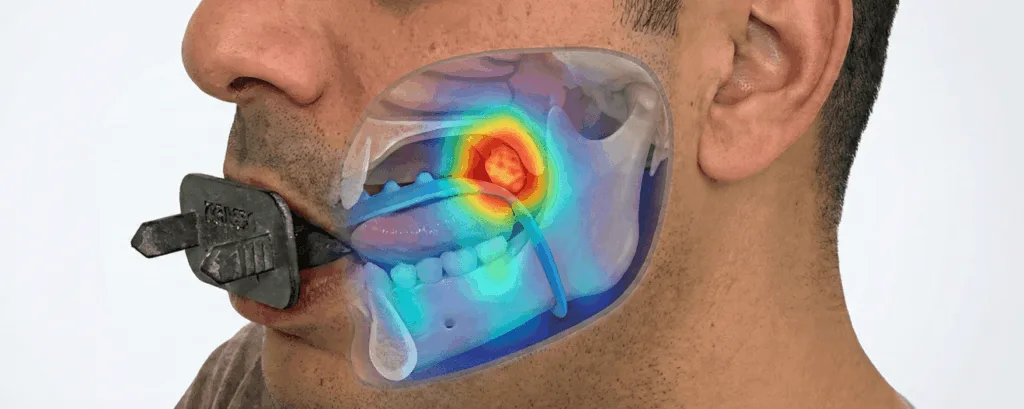
Clinical implementation requires more than just device efficacy. It demands alignment with radiation planning workflows, streamlined fabrication-to-delivery timelines, and acceptable patient tolerance. Kallisio has positioned its technology to meet those demands through a cloud-based ordering portal, rapid five-day scan-to-delivery logistics, and compatibility with standard CT simulation setups.
Stanford’s early adoption suggests that institutions with advanced digital infrastructure and high caseload complexity are likely to lead the curve on integration. But broader diffusion will depend on how well the system performs in terms of reproducibility, therapist workload, and integration with planning software.
Why intraoral stents could become central to head and neck radiation planning
The clinical rationale behind devices like Stentra is well-established. IMRT, now a standard of care in head and neck cancer, relies on sub-millimeter accuracy in dose delivery. Yet, intra-fractional motion, particularly of the tongue, lips, and soft palate, can erode the precision benefits of even the most advanced linear accelerators. A tailored stent that immobilizes oral anatomy offers a low-profile intervention to improve geometric consistency across sessions.
By elevating reproducibility in positioning organs at risk (OARs), Stentra has the potential to shift toxicity management upstream, moving from reactive to preventative. In practical terms, this could mean reduced rates of xerostomia, mucositis, or unintentional brainstem dosing. Clinical observers note that many current positioning techniques fall short in balancing patient comfort with immobilization accuracy, creating an opening for newer, individualized systems.
Importantly, Stentra is not attempting to replace all forms of immobilization but to augment them. Its role is additive, especially useful in cases where the tumor is closely abutting or embedded in mobile oral structures. Radiation oncologists at centers trialing the system are likely to use it selectively, especially in cases involving base-of-tongue tumors, oral cavity malignancies, or re-irradiation protocols.
What this signals about FDA appetite for digitally fabricated, patient-specific devices
The 510(k) clearance of Stentra earlier in 2024 marked a milestone not just for Kallisio, but for the broader class of digitally fabricated, patient-matched therapeutic devices. U.S. Food and Drug Administration clearance for such products has typically hinged on the demonstration of “substantial equivalence” to predicate immobilization systems. However, Kallisio’s integration of intraoral scanning, cloud-based workflows, and rapid 3D printing pushed the boundary of what FDA reviewers now consider compatible with routine clinical use.
Regulatory watchers point out that the agency’s clearance also reflects growing comfort with AI-augmented design processes, even if those processes are not themselves subject to software-as-a-medical-device (SaMD) classification. That distinction will matter if Kallisio expands its platform to incorporate adaptive treatment planning or real-time anatomical modeling. For now, however, the system’s scope remains within the domain of static positioning aids.
The bigger story is the increasing normalization of individualized, digitally manufactured accessories in radiation therapy, especially for anatomically complex regions. As other developers explore similar workflows for thoracic and pelvic sites, Kallisio’s experience could become a template for how to clear, implement, and scale such systems without disrupting existing regulatory or reimbursement frameworks.
What could still limit widespread adoption of Stentra and its platform
Despite its early momentum, Kallisio faces several friction points in driving broader clinical uptake. First, reimbursement codes for patient-specific immobilization devices remain uneven. Unless the company can demonstrate clear cost-offsets through reduced toxicity, shorter setup times, or better dosimetric outcomes, widespread payer adoption could be slow.
Second, the logistical model, which requires intraoral scanning, digital design, and third-party fabrication, introduces additional steps into radiation oncology workflows that are already under staffing pressure. While Kallisio has attempted to streamline this via a cloud-based portal and rapid turnaround times, it will need to prove that this doesn’t create delays or increase time-on-treatment compared to conventional setups.
Clinicians may also hesitate to adopt until more robust data emerges from multi-institutional trials or retrospective studies. Most available insights have come from single-site early use cases, including pilot implementations at academic centers. Without peer-reviewed outcomes data demonstrating consistent improvements in geometric precision, toxicity reduction, or patient-reported outcomes, many community centers may prefer to stick with legacy immobilization strategies.
Manufacturing scalability could also become an issue as adoption grows. Kallisio’s current model appears tailored to U.S. hospital partnerships, but expanding internationally or to high-volume radiotherapy networks would likely require localized production capacity and regulatory approvals in additional jurisdictions.
Why institutional partners like Stanford are critical to future validation
Stanford’s decision to integrate Stentra into routine head and neck cancer workflows lends credibility to the platform and offers a high-profile testing ground for iterative improvements. But the long-term value will lie in whether centers like Stanford publish performance data, particularly on endpoint metrics such as setup reproducibility, dosimetric precision, and clinical toxicity rates.
If early adopters are willing to serve as both validators and co-developers, Kallisio could benefit from a virtuous cycle of feedback-driven product refinement. That might include expanding anatomical coverage, integrating tongue-tracking data, or developing stents for adaptive radiation therapy protocols.
Ultimately, what Kallisio is testing is not just a device, but a new care model: one in which personalization is embedded not only in the treatment plan but in the physical interface between patient and machine. For head and neck cancer patients—where the therapeutic window is narrow and toxicity can be debilitating—that model could be transformative.
What are the key takeaways from Kallisio’s Stentra implementation at Stanford?
- Kallisio’s Stentra system, a 3D-printed patient-specific intraoral device for head and neck cancer radiation therapy, is now in clinical use at Stanford Health Care following its FDA clearance.
- The system targets improved immobilization of oral structures during IMRT, enhancing geometric precision and potentially reducing toxicity.
- Its adoption signals growing comfort among major institutions with digital workflows, cloud-based ordering, and rapid fabrication for individualized accessories.
- Regulatory clearance reflects the U.S. Food and Drug Administration’s evolving stance on digitally manufactured, patient-matched therapeutic devices.
- Reimbursement hurdles, workflow integration challenges, and a lack of long-term clinical outcome data could still limit widespread adoption.
- Stanford’s integration may accelerate validation and real-world data collection, shaping broader acceptance across academic and community centers.