Why Corcept Therapeutics now faces a bigger execution test after Lifyorli’s ROSELLA success

Corcept Therapeutics’ Lifyorli showed strong survival benefit in ovarian cancer. Read what the ROSELLA data could change for clinicians and payers.

Corcept Therapeutics’ Lifyorli showed strong survival benefit in ovarian cancer. Read what the ROSELLA data could change for clinicians and payers.
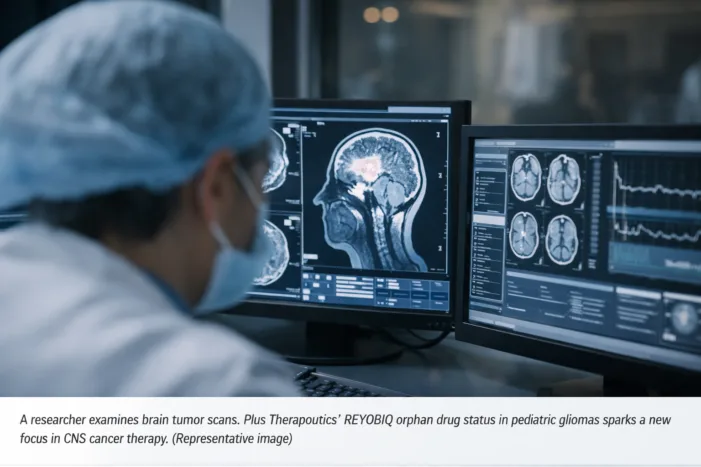
Plus Therapeutics won orphan drug designation for REYOBIQ in pediatric malignant gliomas. Read what it changes for CNS cancer development.

Apotex won first tentative U.S. approval for generic semaglutide. Read what it could mean for glucagon-like peptide-1 access, pricing, and competition.

Lupin Limited has launched generic dapagliflozin in the United States. Read what it means for Farxiga, pricing pressure, and market access.
Eli Lilly and Company’s Foundayo approval could reshape obesity treatment. Read what the oral GLP-1 launch changes for clinicians, payers, and rivals.

Lupin Limited has won tentative FDA approval for pitolisant tablets. Read why timing, exclusivity, and Wakix market dynamics matter next.

AEON Biopharma advances ABP-450 biosimilar strategy. Discover what this means for BOTOX competition and future complex biologics markets.

Phase 1 PK data bring Ameluz closer to broader FDA review. Explore the regulatory stakes and market impact in actinic keratosis.

DifGen Pharmaceuticals secures rare FDA approval for fluorometholone ophthalmic suspension. Find out why this complex generic matters for ophthalmology markets.

Find out how MAIA Biotechnology is advancing ateganosine into Phase 3 and why its 2026 clinical milestones could reshape treatment options in lung cancer.