Abanza Inc. has received U.S. Food and Drug Administration (FDA) 510(k) clearance for QuadLock, its novel knotless fixation system for anterior cruciate ligament (ACL) reconstruction. The system, which features a bidirectional tension-adjustable design, reportedly offers over a 500% reduction in cyclic displacement compared to conventional fixation methods such as cortical buttons and interference screws, targeting enhanced early graft stability during the healing phase.
With this announcement, Abanza joins an increasingly competitive orthopedic landscape, but the question is less about regulatory clearance and more about whether QuadLock’s biomechanical advantages translate to meaningful clinical adoption—and whether the company’s platform strategy can sustain traction in high-demand procedural segments like ACL repair.
What QuadLock’s mechanics reveal about surgical priorities in modern ACL reconstruction
The most noteworthy specification in Abanza’s QuadLock system is its capacity to maintain cyclic displacement under 0.5 mm under repeated loading—an outcome that significantly outperforms the 3–6 mm displacement typically observed with traditional methods. This tight control may reduce the risk of graft elongation and instability, which is especially relevant in the immediate post-operative period when fixation integrity can determine patient outcomes.
By delivering >1,000 N pullout strength alongside knotless tension adjustability, the system appears to directly address one of the central trade-offs in current fixation solutions: the balance between ease of use, secure fixation, and intraoperative flexibility across varied graft types and bone qualities.
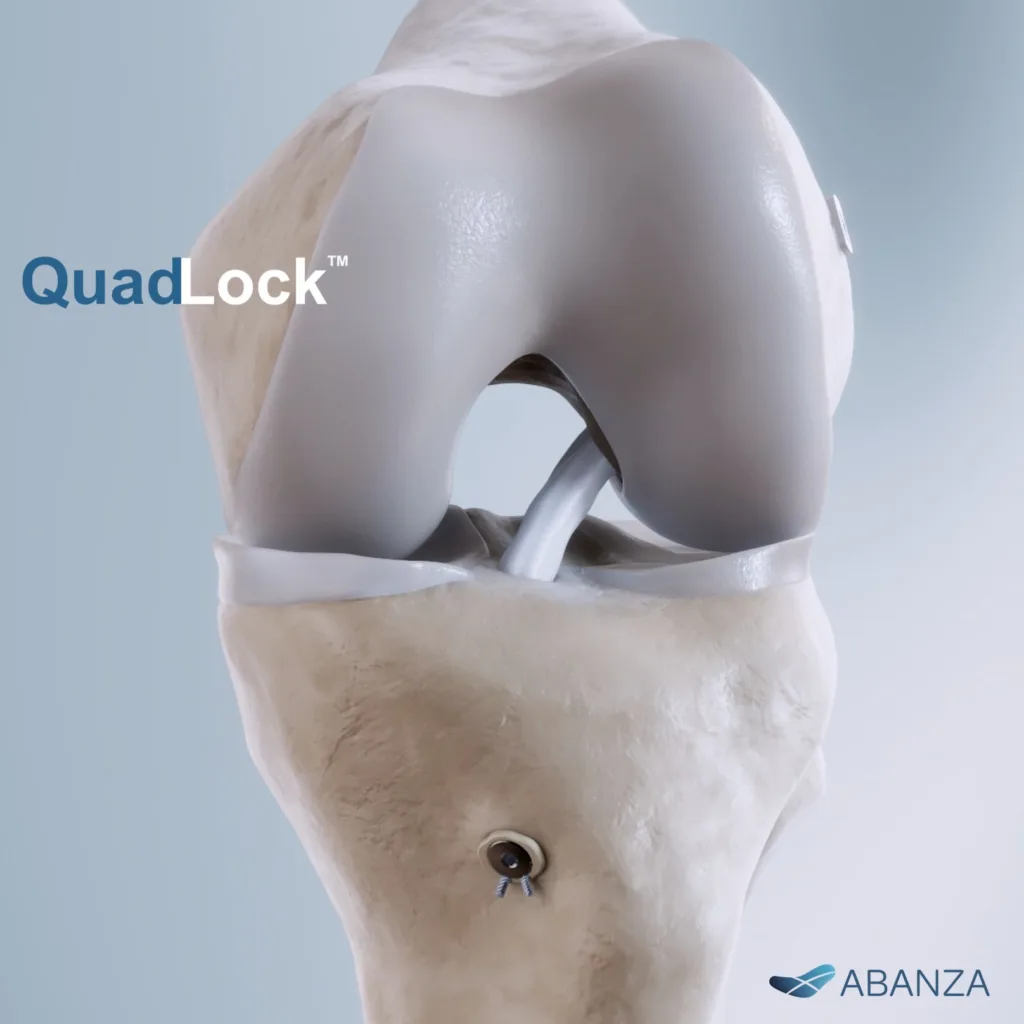
The bidirectional nature of QuadLock’s tensioning system could offer surgeons more real-time control when positioning quadriceps tendon, hamstring, or bone–patellar tendon–bone grafts—especially in patients with less predictable bone density, such as older athletes or adolescents. But while these features are theoretically sound, adoption will hinge on how they perform under real surgical pressures, not just in biomechanical test rigs.
Why procedural reproducibility is becoming the next battleground in fixation systems
Industry observers tracking orthopedic innovation note that surgical reproducibility is rapidly emerging as the next major differentiator—not just peak performance in lab conditions. Devices that offer consistent outcomes with lower variability across skill levels and patient anatomies are more likely to gain favor, especially in high-volume sports medicine practices.
QuadLock’s knotless approach and pre-assembled delivery configuration aim to reduce intraoperative complexity. Combined with the tension-adjustable capability, the system may allow more predictable graft placement and potentially reduce revision rates stemming from poor initial tensioning. However, the absence of peer-reviewed comparative data or early clinical case series will be a bottleneck for widespread clinical uptake.
Clinicians are likely to want to see not only proof of superior mechanical metrics but also evidence that the implant can be deployed efficiently across surgical centers with varied resources. Early adopters will probably evaluate this through hospital-based pilot deployments before recommending it for routine use.
What this approval signals about Abanza’s broader platform strategy and commercial runway
The FDA nod for QuadLock gives Abanza a legitimate foothold in the crowded ACL reconstruction device market, which is dominated by incumbents such as Arthrex, Smith & Nephew, and Zimmer Biomet. Rather than attacking these players directly, Abanza appears to be pursuing a portfolio-based approach, anchoring its commercial push on mechanical strength and reproducibility, while expanding its applications horizontally into adjacent joints and tendons.
With additional devices like LoopCap and WasherCap In Line in development for biceps tendon repair, AC joint reconstruction, and Achilles fixation, the company is clearly positioning itself as a multi-joint soft-tissue fixation platform rather than a single-product challenger.
But platform potential will be constrained by whether Abanza can show durable advantages across diverse tissue types and fixation geometries. The leap from femoral tunnels in ACL procedures to AC joint or syndesmosis repair is not trivial in terms of load paths, soft tissue interface, or surgeon technique. The mechanical claims will need to be revalidated in each use-case context.
What regulatory clarity exists—and what remains uncertain for scale-up and reimbursement
FDA 510(k) clearance is a necessary but not sufficient condition for commercial success. In QuadLock’s case, clearance was likely based on substantial equivalence to existing suture-based or adjustable button fixation systems, which means the bar for demonstrating safety and performance was technical, not clinical.
This pathway reduces time to market but doesn’t confer any specific coding or reimbursement advantage. Without a unique CPT code or documented procedural efficiency gains, adoption will depend largely on the device’s perceived value to the surgeon, not the billing department.
Additionally, 510(k) devices often face a fragmented payer landscape, especially in outpatient ambulatory surgery centers. To address this, companies in similar positions have often pursued registry inclusion or postmarket surveillance studies to build an economic value case—a route Abanza may need to consider if it seeks hospital formulary traction or bundled payment inclusion.
What could go wrong: risks in real-world deployment and competitive positioning
While QuadLock’s performance metrics are impressive in controlled environments, the primary risks lie in surgical translation. If the tension-adjustable mechanism introduces complexity or malfunctions under real-use stress, early reputation damage could be significant.
Moreover, tension control in ACL reconstruction is as much an art as a science. Surgeons accustomed to fixed-point devices or screw-based fixation may not immediately perceive a compelling need to switch, especially without robust training programs or side-by-side evidence in clinical series.
Competitively, the device must also prove its relevance beyond high-performance centers. If QuadLock is perceived as a “premium” solution for niche scenarios rather than a dependable mainstay across routine procedures, its market ceiling may remain low.
And while Abanza’s broader strategy of building a fixation platform is compelling, success depends on consistent performance across every anatomic indication it targets. A misstep in a secondary joint—say, Achilles or MPFL—could undermine confidence in the core ACL use case.
What this 510(k) clearance enables—and why scrutiny will intensify post-launch
Abanza’s QuadLock clearance establishes a new reference point in biomechanical performance metrics for ACL fixation—but performance alone won’t secure commercial dominance. Clinicians, procurement teams, and health systems will be watching for proof that the device delivers not just superior strength, but superior predictability, reproducibility, and ease of use.
The next phase of scrutiny will likely focus on: time-to-graft incorporation, intraoperative deployment time, failure-to-fixation incidents, and comparative clinical outcome data. These will shape not just whether the device is adopted, but whether it becomes central to soft-tissue fixation strategy or remains peripheral.
In a procedural domain where trust and habit weigh heavily, QuadLock’s bidirectional innovation now faces its real test—in the hands of surgeons under the lights of the OR.