Veradermics Incorporated has completed enrollment in its pivotal Phase 2/3 clinical trial for VDPHL01, a novel extended-release oral minoxidil formulation designed for the treatment of male pattern hair loss. The late-stage biopharmaceutical company has also initiated a second Phase 3 trial in men and a Phase 2/3 trial in women, positioning VDPHL01 as a potential first-in-class, non-hormonal oral therapeutic in a dermatology space long dominated by hormonal agents and off-label solutions. If successful, the program could break a 30-year drought in new FDA-approved prescription treatments for androgenetic alopecia.
Why an extended-release oral minoxidil could mark a turning point in alopecia care
Unlike topical minoxidil products or immediate-release oral formulations used off-label, VDPHL01 is specifically engineered for follicular targeting with systemic safety in mind. Veradermics Incorporated has designed the tablet to gradually release minoxidil over time, ensuring consistent blood concentrations that remain below cardiac risk thresholds while prolonging exposure at the scalp level.
This pharmacokinetic strategy aims to address a longstanding issue with oral minoxidil. While dermatologists have reported encouraging results from off-label use, concerns around sudden peak concentrations, especially in patients with cardiovascular vulnerabilities, have kept it outside the bounds of mainstream prescription care. The gel-matrix extended-release design in VDPHL01 attempts to thread this needle by maintaining therapeutic levels that foster hair regrowth without triggering adverse cardiovascular events.
In this context, Veradermics Incorporated is positioning VDPHL01 not as a repurposed cardiovascular drug, but as a purpose-built dermatology asset for the largest aesthetics indication in the world.
What differentiates VDPHL01 from finasteride and other legacy treatments
Currently, the only oral prescription medication approved for male pattern hair loss is finasteride, a 5-alpha-reductase inhibitor that works by suppressing the conversion of testosterone to dihydrotestosterone. While effective for many, finasteride’s hormonal mechanism introduces a range of tolerability and safety concerns, particularly regarding sexual, reproductive, and psychological side effects. Moreover, the drug is not recommended for use in women, especially those of childbearing potential, creating a significant treatment gap in female androgenetic alopecia.
VDPHL01 bypasses the hormonal axis altogether. Its mechanism relies on vasodilation and stimulation of follicular activity through non-endocrine pathways. This could potentially make it suitable for a broader patient population, including women, pending favorable safety data from the ongoing female Phase 2/3 trial. Importantly, this non-hormonal approach reduces the stigma and physiological risk factors often associated with finasteride therapy, especially among younger patients.
From a clinical differentiation standpoint, dermatologists tracking the field have noted that if Veradermics Incorporated can demonstrate comparable or superior efficacy to finasteride with a better safety profile and broader applicability, it could become a go-to first-line option. It may even serve as a dual-pathway partner in future combination regimens targeting both hormonal and non-hormonal drivers of hair loss.
What early data shows—and what the FDA will want to see next
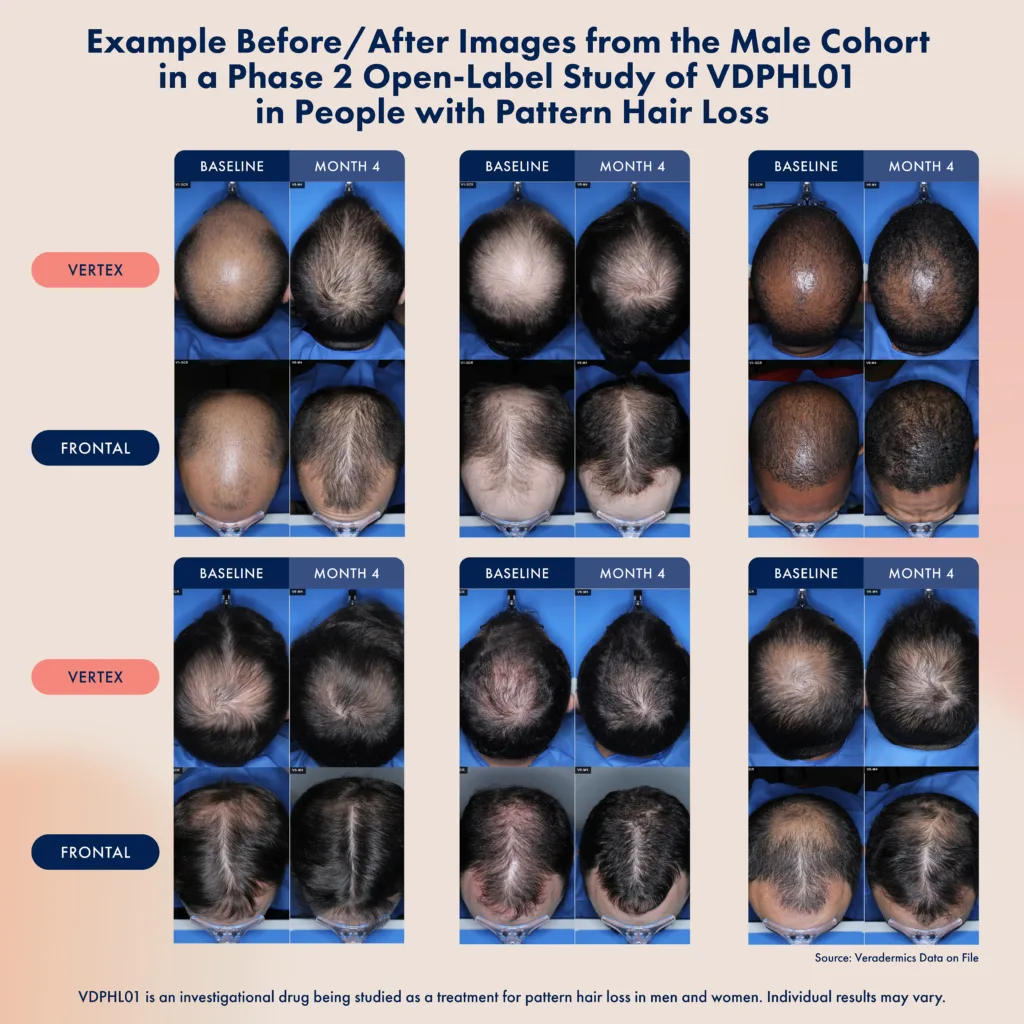
The current Phase 2/3 trial enrolled 519 male participants with mild to moderate pattern hair loss, randomized to receive either 8.5 mg of VDPHL01 once daily, twice daily, or placebo over a 52-week period. The primary endpoints include changes in non-vellus hair count and patient-reported hair coverage benefit at 24 weeks. While topline results are not yet available, Veradermics Incorporated has shared preliminary data from a separate Phase 2 cohort that appears encouraging.
According to interim data, participants experienced an average gain of 47.3 hairs per square centimeter at four months, with noticeable gains of 37.5 hairs per square centimeter as early as two months. Patient-reported outcomes also appear strong, with 55 percent of subjects reporting visible improvement at two months and over 90 percent reporting improvement by four months. Additionally, VDPHL01 was reportedly well tolerated, with no serious adverse events, including no cardiac-related side effects.
While these signals are promising, regulatory approval will hinge on consistent performance in the registration-directed study, particularly around cardiac safety, which remains a known risk with systemically absorbed minoxidil. Investigators will need to show that the extended-release format can sustain therapeutic benefits while avoiding arrhythmias, hypotension, or other cardiovascular complications.
Why the gender split in development could reshape the entire treatment landscape
Pattern hair loss is not exclusive to men. An estimated 30 million women in the United States are affected by androgenetic alopecia, representing a massive unmet need in dermatologic therapeutics. However, most approved prescription therapies have either been male-focused or carry contraindications in women, particularly during reproductive years.
By developing VDPHL01 in parallel male and female cohorts, Veradermics Incorporated is pursuing one of the few clinical programs that could result in a single, non-hormonal oral therapy approved across both populations. If the female cohort reproduces the efficacy and safety seen in male trials, the company may secure a unique position in the marketplace as the only broadly prescribable oral agent for pattern hair loss.
Clinicians and regulatory watchers alike will be focused on the female trial’s outcomes, especially considering the absence of hormonally driven safety concerns, which may allow for a differentiated label compared to finasteride or spironolactone.
What market dynamics reveal about prescription opportunity and unmet demand
Industry observers suggest that the long stagnation in FDA approvals for pattern hair loss—now approaching three decades—has led to a fragmented and often unregulated market. While over-the-counter products and “nutraceuticals” remain popular, their inconsistent efficacy and lack of clinical validation have limited dermatologist confidence and patient satisfaction. This has created an unusual dichotomy where consumer demand is sky-high, but few prescription options exist to meet it.
The market size reflects this paradox. According to industry estimates, the global pattern hair loss market is expected to exceed 30 billion dollars by 2028, driven by aesthetic demand, an aging population, and increasing societal acceptance of hair loss treatment. Within this space, an FDA-approved, prescription-strength oral minoxidil could quickly gain traction—particularly if it earns payer coverage and is priced competitively against compounded alternatives.
However, commercialization success will also depend on Veradermics Incorporated’s ability to educate prescribers, particularly primary care physicians and non-dermatology specialists who may be unaware of the evolving oral treatment landscape. The extended-release format could also help differentiate VDPHL01 from low-cost compounded generics, giving it a premium positioning supported by clinical data and regulatory oversight.
What risks remain before VDPHL01 can shift the treatment standard
Despite the promise, VDPHL01 is not yet a guaranteed success. The extended-release design, while innovative, introduces complexity in manufacturing and consistency that must hold up across large-scale production. Even minor variations in release kinetics could impact safety and efficacy, which regulators will likely scrutinize closely.
Additionally, the placebo effect in dermatologic aesthetics is notoriously high. Subjective endpoints like patient satisfaction or hair coverage perception must be balanced with objective measures like hair count and follicular density. The FDA will want to see statistical significance across both domains before moving toward approval.
Payers may also be hesitant to reimburse a therapy seen primarily as cosmetic, unless Veradermics Incorporated can position it within the broader context of quality-of-life impact, including links to mental health and self-esteem documented in alopecia literature. Without reimbursement, even a successful approval could face limited adoption among cost-conscious patients.
What the next milestones signal for dermatology and aesthetics innovation
The next 12 to 18 months will be pivotal. Full data from both male and female trials is expected in 2026. If results hold, Veradermics Incorporated could file for regulatory approval shortly thereafter, potentially becoming the first company in decades to secure an FDA nod for a new oral hair loss drug.
More broadly, the company’s approach reflects a shifting trend in dermatology, where common but historically underprioritized conditions like alopecia, rosacea, or hyperpigmentation are being re-examined through the lens of serious medical burden and regulatory legitimacy. If VDPHL01 succeeds, it could catalyze new interest from biopharmaceutical developers to enter the aesthetics space with rigorously developed, FDA-approved therapeutics.